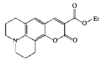
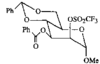
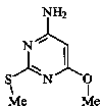
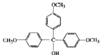
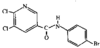
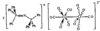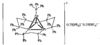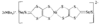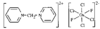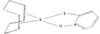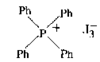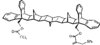issue contents
February 1996 issue
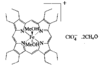
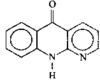
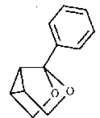
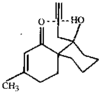
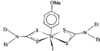
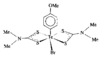
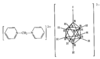
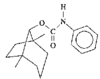
Cover illustration: A 5,18:9,14-Di-o-benzeno-6,17:7,16:8,15-trimethanobenzo[1'',2'':3,4;4'',5'':3',4']dicyclobuta[1,2-b:1',2'-b']dianthracene Derivative, see Parvez pages 444-447. Displacement ellipsoids are plotted at the 50% probability level.
inorganic compounds
Download citation


Download citation


Rb2[UO2Cl4].2H2O crystallizes in space group P1; the anion approximates to D4h (4/mmm) symmetry, with a U—O distance of 1.751(9)Å and U—Cl distances of 2.644(3) and 2.650(4)Å. The compound is confirmed to be the dihydrate; the degree of hydration had been controversial.
Download citation


Download citation


The structure of Re6Te4Cl10 contains two independent Re6 octahedral clusters, each with a crystallographic center of symmetry. The Re atoms are coordinated by four μ3-Te atoms and four μ3-Cl atoms that are equally distributed over the faces of the metal polyhedron; the remaining six Cl atoms are terminal ligands coordinated to the apices of the Re6 octahedron.
Download citation


Download citation


The structure of K2Mn(VO3)4 is composed of MnO6 octahedra and VO4 tetrahedra. Parallel zigzag polyvanadate (VO3)nn- chains of corner-sharing VO4 tetrahedra are cross-linked by seperate MnO6 octahedra to form a three-dimensional framework, the cavities of which are occupied by K+ ions.
Download citation


Download citation


The structure of K2Mn3(OH)2(VO4)2 consists of [Mn3(OH)2(VO4)2]n2n- layers built up from CdI2-like planes of MnO4-(OH)2 octahedra with 1/4 Mn vacancies, to which VO4 tetrahedra are linked on both sides via three vertices; the fourth vertex points into the interlayer space where the K+ ions are situated.
Download citation


Download citation


The Sn6O4(OH)4 clusters which form the structure pack in a pseudo-body-centered fashion. The Sn atoms have distorted tetragonal pyramidal coordination geometry; each Sn atom is coordinated to two bridging O atoms and two bridging hydroxy O atoms.
Download citation


Download citation


The structure of sodium hexanitroruthenate(II) monohydrate is composed of [Ru(NO2)6]4− complex anions, Na+ cations and water molecules. The Ru atoms have nearly ideal octahedral coordination geometry.
Download citation


Download citation


The structure of P2I4 was determined at 120K. It is composed of isolated P2I4 molecules, each of which lies on a crystallographic symmetry centre. There are no apparent intermolecular contacts between the molecules.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


addenda and errata
Free 

Erratum to Acta Cryst. (1994), C50, 984-986.


 journal menu
journal menu