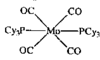
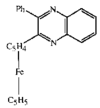
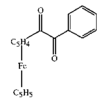
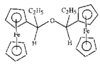
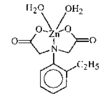
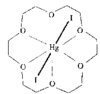
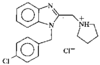
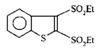
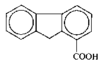
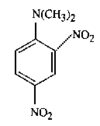
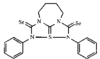
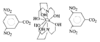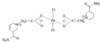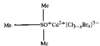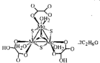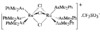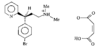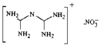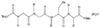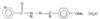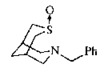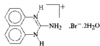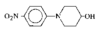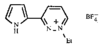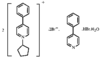issue contents
April 1996 issue
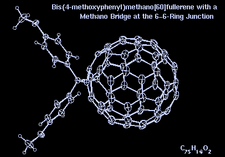
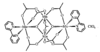
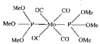
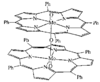
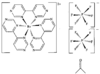
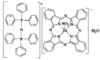
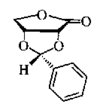
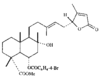
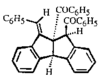
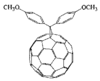
Cover illustration: Bis(4-methoxyphenyl)methano[60]fullerene with a Methano Bridge at the 6-6-Ring Junction, see Tezuka, Kawasaki, Yajima, Ishii, Oyama, Takeuchi, Nakao & Katayama, pages 1008-1010. Displacement ellipsoids are plotted at the 30% probability level.
inorganic compounds
Download citation


Download citation


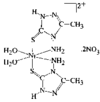
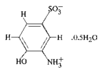
The Al atoms in Na9[Al(OH)6]2(OH)3.6H2O have octahedral coordination geometry and exist as monomeric [Al(OH)6]3− anions. The Na+ ions also have octahedral coordination geometry and are coordinated by OH− ions and water molecules. The NaO6 octahedra are linked through common edges, incorporating the [Al(OH)6]3− octahedra, to yield a three-dimensional network.
Download citation


Download citation


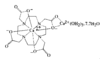
The orthophosphate Cs8Fe15F10(PO4)12 is built from layers of vertex-sharing Fe polyhedra linked into a framework by PO4 tetrahedra. The Fe3+ ions have octahedral coordination geometry and the Fe2+ ions have octahedral, tetrahedral and distorted tetragonal pyramidal coordination environments.
Download citation


Download citation


The three-dimensional polymeric complexes [Bi(SCN)6La(H2O)3].2H2O and [Bi(SCN)6Nd(H2O)3].2H2O are isomorphic with and have a similar structure to the LaFe(CN)6.5H2O structure type. The bond lengths in the coordination polyhedra and the hydrogen bonding in the structures are discussed.
Download citation


Download citation


Cs8[SiW10O36].9H2O contains the heteropolyoxometalate anion γ-[SiW10O36]8−. The polyanions are connected by Cs+ ions to form infinite columns.
Download citation


Download citation


Barium gallium nitride, Ba3Ga2N4, is isostructural with Sr3Al2N4. The structure contains GaN4 tetrahedra which share edges to form infinite [GaN4/23−] chains.
Download citation


Download citation


The structure of Co2Si(P2O7)2 contains SiO4 tetrahedra, eclipsed P2O7 groups [with a P-O-P angle of 132.3(1)°] and compressed CoO6 octahedra. The CoO6 octahedra share edges to form Co2O10 units, which are linked via four vertices to form a two-dimensional network.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu