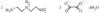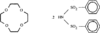issue contents
June 1998 issue
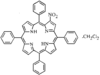
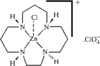
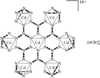
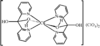
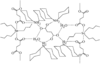
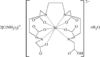
Cover illustration: Star-Shaped Heptanuclear Tetramine Cadmium(II) Complex, see Klüfers & Mayer, pages 722-725. Displacement ellipsoids are shown at the 60% probability level for non-H atoms and perchlorate molecules have been omitted for clarity.
cif-access (inorganic compounds)
Download citation


Download citation


RbCe(SO4)2.H2O has a framework structure composed of discrete sulfate tetrahedra interlinked by nine-coordinate Ce polyhedra. The framework contains large channels in which the Rb atoms reside, in contrast to the layered structure exhibited by the analogous tetrahydrate.
cif-access (organic compounds)
Download citation


Download citation


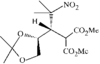
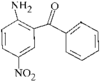
The porphyrin molecule in this compound shows significant conformational distortion in the vicinity of the beta-nitro group. This is attributed to steric interaction between a meso-phenyl group and the beta-nitro group.
Download citation


Download citation


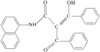
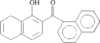
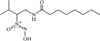
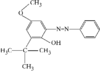
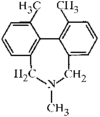
The phenyl rings in this compound form a dihedral angle of 46.8(1)° and they make dihedral angles of 79.6(1) and 67.7(1)° with the naphthalene ring. The hydroxy groups are involved in O-H⋯O intramolecular hydrogen bonds.
Download citation


Download citation


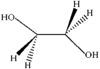
Ethylene glycol adopts a synclinal conformation in the solid state. The molecules pack to form a two-dimensional network with four hydrogen bridges per molecule [O⋯O 2.711(1) and 2.730(2)Å].
Download citation


Download citation


Very different hydrogen-bonded networks are found in the two forms. The monoclinic form (in which the cation is trans-trans) has open fused columns and a unit-cell volume 5% greater than the triclinic form (in which the cation is trans-gauche), where the network contains fused clusters.
inorganic compounds
Download citation


Download citation


The structure of Nd2(Se2O5)3.H2SeO3.2H2O is built up from polar layers formed by Nd2O16 bipolyhedra and diselenite groups. Water and selenious acid occur on only one side of each layer.
Download citation


Download citation


Magnesium bis(dihydrogenorthophosphate) tetrahydrate contains four-membered rings made up of two [PO4] tetrahedra and two [MgO6] octahedra sharing corners.
Download citation


Download citation


The different colours of turquoise Cs3NiCl5 and orange CsNiCl3 are caused by the tetrahedral coordination of nickel(II) in the former and the octahedral coordination of nickel(II) in the latter.
Download citation


Download citation


Rb4Ta2S11 and Cs4Ta2S11 are new members of the growing family of compounds which contain an M2Q11 unit as a main structural feature. They were prepared via the reactive flux method.
Download citation


Download citation


Mo6Te8, isotypic with Mo6Se8, contains interlinked Mo6Te8 clusters. The eight faces of each distorted Mo6 octahedron are capped by Te atoms, six of which provide bridges to neighboring Mo6Te8 clusters.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


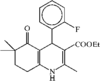
organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


addenda and errata
Download citation


Download citation


Free 

Addendum to Acta Cryst. (1991), C47, 2659-2660.
international union of crystallography
Free 



 journal menu
journal menu