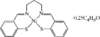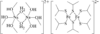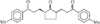issue contents
July 1999 issue
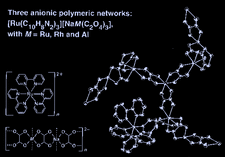
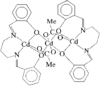
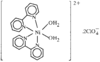
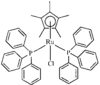
Cover illustration: Three anionic polymeric networks: sodium(I)-ruthenium(III), sodium(I)-rhodium(III) and sodium(I)-aluminium(III) mu-oxalato complexes with tris(2,2'-bipyridine)ruthenium(II) cations, see Pellaux, Decurtins & Schmalle, pages 1075-1079. Displacement ellipsoids are shown at the 50% probability level.
cif-access (inorganic compounds)
Download citation


Download citation


Low-temperature potassium iron germanium tetraoxide is a stuffed tridymite derivative with a completely disordered Fe/Ge tetrahedral distribution.
Download citation


Download citation


The low-temperature β-KFeSiO4 (potassium iron silicate) polymorph crystallizes with a kalsilite-type structure having P6322 symmetry and a disordered Fe/Si tetrahedral distribution.
cif-access (organic compounds)
Download citation


Download citation


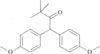
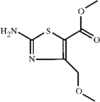
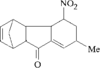
The title compound was obtained as a by-product of the reaction of pinacolone and 4-iodoanisole. The crystal structure was determined at 120(2)K. The molecule is bisected by a mirror plane (space group Pnma).
Download citation


Download citation


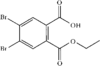
The molecular structure of 4,5-dibromophthalic acid monoethyl ester is normal. The carboxylic acid group is rotated 12.8° and the ethoxycarbonyl group 69.0° out of the mean plane of the benzene ring.
Download citation


Download citation


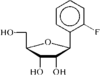
The ribofuranose ring in the title compound has a C2'-endo envelope conformation. The crystal packing consists of intermolecular hydrogen-bonding and phenyl⋯phenyl interactions.
Download citation


Download citation


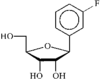
The structure of the title compound is isostructural with 1-deoxy-1-phenyl-β-D-ribofuranose [Matulic-Adamic et al. (1996). J. Org. Chem. 61, 3909–3911]. The ribofuranose ring has a C2'-endo,C3'-exo twist conformation.
Download citation


Download citation


The two crystallographically independent conformations of the title compound differ only by the rotational positions adopted by their methoxymethyl substituents. Intermolecular contacts are dominated by hydrogen bonding involving the amine H atoms.
Download citation


Download citation


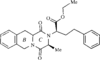
The title compound, ethyl 2-(1,2,3,4,5,6,11,11a-octahydro-3- methyl-1\,4-dioxopyrazino[1\,2-b]isoquinolin-2-yl)-4-phenylbutanoate, is formed due to cyclization of quinapril in solution. The configurations at the three chiral centres are all S. The six-membered B and C rings assume half-chair and boat confirmations, respectively. The molecules in the lattice are stabilized by van der Waals contacts.
Download citation


Download citation


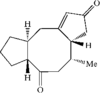
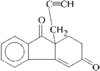
The title compounds, C15H20O2, (I), and C15H22O3.CH3OH, (II), were obtained by intramolecular cyclization of the 5- and 3-acetonyl derivatives of 4-methylbicyclo[6.3.0]undecane-2,6-dione, respectively. Dione (I) is an ophiobilin-like C5–C8–C5 fused-ring compound and diol (II) is a C5–C6–C5–C5 tetracyclic compound.
Download citation


Download citation


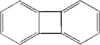
Biphenylene is one of the key compounds in the discussion of bond localization in aromatic systems, usually addressed as the Mills–Nixon effect [Mills & Nixon (1930). J. Chem. Soc. p. 2510]. A pronounced bond localization due to the Mills–Nixon effect in the direction of one of the Kekule forms is established. Compared with the previous structure determination by Fawcett & Trotter [Acta Cryst. (1966), 20, 87-93], the significance of bond-length alternation in relation to the s.u.'s in the six-membered rings has been dramatically increased.
Download citation


Download citation


The title compound crystallizes with two nearly identical molecules in the asymmetric unit. The molecules essentially exhibit mirror symmetry.
Download citation


Download citation


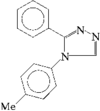
In the title compounds, C10H8C12N4 (R= H), (I), and C18H20C12N4O2 (R = COCHMe2), (II), the dihedral angle between the imidazolidine and phenyl rings are 67.7(1) and 70.34(9)°, respectively. In (I), the imidazolidine ring adopts a half-chair conformation, whereas in (II), it is in a flattened envelope conformation.
inorganic compounds
Download citation


Download citation


The structure of Na2Ca2Nb4O13 consists of perovskite-type layers with a thickness corresponding to four perovskite cubes. The interlayer structure is of the rock-salt-type, composed of Na and O atoms. The atomic configuration suggests a ferroelectric nature along the b axis.
Download citation


Download citation


β-Ba5Sb4 displays a more symmetrical arrangement than α-Ba5Sb4 (Pnma). It contains two anionic moieties, Sb24− and Sb3−, lying in tricapped and distorted monocapped trigonal prisms of Ba atoms, respectively.
Download citation


Download citation


The reaction of K2S3, tellurium and sulfur yields light-yellow single crystals of dipotassium tellurium trisulfide, K2TeS3. The structure consists of isolated TeS32− anions, and K+ cations coordinated by six S atoms from five different TeS32− anions within distorted octahedra.
Download citation


Download citation


KNa(VO3)2 adopts the pyroxene structure with K+ and Na+ cations, which lie on twofold axes, occupying the dodecahedral and octahedral cavities, respectively.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu