issue contents
October 2000 issue
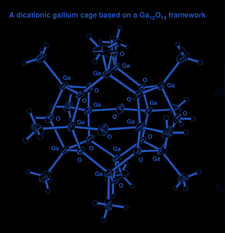
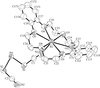
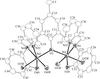
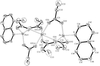
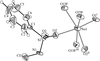
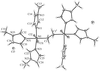
Cover illustration: A dicationic gallium-oxo-hydroxide cage compound. Displacement ellipsoids are shown at the 35% probability level. See Swenson, Dagorne & Jordan [Acta Cryst. (2000), C56, 1213-1215].
inorganic compounds
Download citation


Download citation


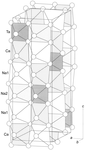
The title quaternary oxide, trisodium dicalcium tantalum hexaoxide, is isostructural with Li3Ni2TaO6, a partially ordered rock-salt phase. The Na, Ca and Ta atoms occupy octahedral sites in an orderly manner and form a cation-ordered superstructure.
Download citation


Download citation


A full single-crystal structure analysis of [Ba(OH)I(H2O)4] reveals intra- and intermolecular hydrogen bonding, leading to a three-dimensional network. The compound is an intermediate between BaI2 and the gel of Ba(OH)2(H2O)x.
Download citation


Download citation


The structure of K3NaSn3Se8 is composed of tritetrahedral [Sn3Se8]4− anions, which are separated by four-coordinate Na+ and eight-coordinate K+ cations.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


electronic papers (inorganic compounds)
Download citation


Download citation


Synthetic mansfieldite is confirmed to be a variscite-type phase. cis-AlO4(H2O)2 octahedra and AsO4 tetrahedra form a dense vertex-sharing network. The O—H⋯O hydrogen-bonding network has been elucidated.
Download citation


Download citation


Caesium vanadium(V) selenite contains vertex-sharing layers of distorted VO6 octahedra, capped by pyramidal selenite groups. Interlayer caesium cations complete the structure, which is isostructural with M(VO2)3(SeO3)2 (M = NH4+, K+) and Cs(VO2)3(TeO3)2.
Download citation


Download citation


The title double salt was obtained from the reaction of Y(NO3)3 and K6[H4Co2Mo10O38]·5H2O at a pH of about 2.0. The [H6CoMo6O24]3− anion is a typical B-type Anderson-structure heteropolyanion.
electronic papers (metal-organic compounds)
Download citation


Download citation


The structure of the title compound comprises a zigzag polymer of alternating tetrakis(μ-carboxylato-O:O′)dicopper(II) and 2-aminopyrimidine units linked by axial Cu—N bonds, and the non-centrosymmetric structure has four unique (3-methoxyphenyl)acetate moieties.
Download citation


Download citation


In complexes of the ligand 3,5-bis(2-pyridinyl)-1,3,4-oxadiazole (pod), coordination is through one pyridine and one oxadiazole N atom, i.e. it behaves as a bidentate ligand. Octahedral coordination of the Cu or Ni metal atom in the title complexes, (I) and (II), respectively, is achieved by two pod ligands and two water molecules in axial positions.
Download citation


Download citation


The title ruthenium complex contains a monodentate (C4H9)PPh2 ligand coordinated by the P atom. Coordination about the metal centre is completed by a η6-p-cymene ligand and two Cl atoms.
Download citation


Download citation


Bridging by the two thiocyanate groups in centrosymmetric six-coordinate bis(diphenylthiourea)bis(thiocyanato)cadmium(II) dihydrate leads to the formation of eight-membered [Cd—SCN→Cd—SCN→] rings that are linked at the metal atom to furnish chains running parallel to the a axis.
Download citation


Download citation


In the title compound, the sodium ion is coordinated in a distorted octahedral manner by two carboxylate O atoms and two water O atoms. Each of these water molecules bridges two adjacent Na ions, resulting in two four-membered rings of the type Na–O–Na–O.
Download citation


Download citation


A pseudo-tetrahedral Ni complex has been formed as a result of incomplete bis-chelation of Ni2+ by L−. The dihedral angle at nickel is 68.8 (1)°. A trigonal–planar LiO3 coordination is formed, with Li—O distances of 1.801 (6), 1.825 (6) and 1.842 (6) Å.
Download citation


Download citation


The three title complexes, (I)–(III), are isomorphous and isostructural and have crystallographic D3 symmetry. The lanthanide metal ions are coordinated by nine O atoms in a skewed tricapped trigonal prismatic geometry.
Download citation


Download citation


Reaction of CuBr2 with 2,6-diaminopyridine in HBr solution leads to 3,5-dibromination of the pyridininium ring. The resultant cation was isolated as the [CuBr4]2− salt. The diaminodibromopyridinium ions are planar, while the CuII ions have a distorted compressed tetrahedral coordination with C2 symmetry.
Download citation


Download citation


The structure consists of C7H11N2+ cations and centrosymmetric [SnCl6]2− octahedra. The octahedral [SnCl6]2− anion is hydrogen bonded via trans-Cl atoms to pyridinium N atoms from two cations forming (C7H11N2)2[SnCl6] structural units.
Download citation


Download citation


The first RuIII–thz (thz is 1,3-thiazole) complex characterized via X-ray diffraction, mer-[RuCl3(C3H3NS)3], is a potentially cytostatic compound and consists of discrete complex molecules with an octahedral coordination sphere in which the metal centre is linked to three chloride ions and to three thz ligands through the N atoms. The compound was prepared from the reaction of K3RuCl6 and excess thiazole in MeOH. The Ru—Cl and Ru—N bond distances average 2.3462 (6) and 2.0851 (19) Å, respectively.
Download citation


Download citation


The structure of the title compound consists of β-[Mo8O26]4− polyanions and imidazolium cations. A γ→β conversion is produced in the presence of Cu(NO3)2·3H2O.
Download citation


Download citation


The centrosymmetric [Mo4O10(OCH3)4Cl2]2− anion is built up of four edge-sharing octahedra, two MoO6 and two MoO5Cl.
Download citation


Download citation


In the title complex, the Mn atom is coordinated by three N atoms and three O atoms from three pyridinecarboxylate ligands in a distorted octahedral geometry. One ligand is protonated and is hydrogen bonded to the hydroxide anion.
Download citation


Download citation


The title compound is a one-dimensional heterometallic coordination polymer of the zigzag-type with the molecular composition [(dmpda)Pt(isonic)2Cu(H2O)3](NO3)2, where dmpda is 2,2-dimethyl-1,3-propanediamine and isonic is isonicotinate. One Cu atom links two platinum complex molecules through coordination to two carboxylate groups from the two platinum complex units, and the polymer has repeat zigzag units in which the Cu—Pt—Cu angle is 64.9°.
Download citation


Download citation


The Fe atom in the low-temperature (173 K) structure of the title metal–organic chemical vapour deposition (MOCVD) precursor is octahedrally coordinated and the three chelate rings are found to be significantly non-planar, adopting a half-chair conformation with the Fe atom out of the plane. The structure exhibits only intramolecular C—H⋯O interactions.
Download citation


Download citation


The title compound is found to exhibit a distorted trigonal–bipyramidal geometry at both SnIV atoms. The Sn—Cl—Sn angle involving the bridging chlorine ligand is 135.56 (5)°. The values of the Sn—Cl bond lengths in the bridge are 2.5704 (13) and 3.1159 (13) Å.
Download citation


Download citation


The crystal structure of the title compound shows a distorted octahedral geometry for the Sn atom. As in 1:2 complexes of dimethyltin dihalides with N-methylpyrrolidinone (NMP), the coordination of the amide ligand to SnIV is realised via oxygen.
Download citation


Download citation


The NiII ion attains a trans-octahedral coordination with two bidentate phenylenediamine ligands and two chloride ions in the coordination sphere.
electronic papers (organic compounds)
Download citation


Download citation


The title compound has a central double-bond length of 1.3694 (12) Å. The two benzene rings, lying on different sides of the 2-butenedioate main chain, are almost parallel to each other and nearly perpendicular to the main chain.
Download citation


Download citation


The title compound is composed of a benzodiazepine fragment with a five-membered triazole ring fused on a seven-membered ring. The triazole ring, which has a nitrophenyl substituent, is planar and slightly twisted with respect to the nitrophenyl ring.
Download citation


Download citation


The title compound is composed of a benzodiazepinone fragment with two five-membered rings (oxadiazole and cyclopentane) fused to the seven-membered diazepine ring. The oxadiazole ring, which bears a mesityl group, has a bipyramidal conformation, while the cyclopentane ring has an envelope conformation.
Download citation


Download citation


The title compound was obtained by condensation between benzodiazepine and diethyl 2,3-epoxy-1,4-butanedioate. An oxazoline ring is fused on the benzodiazepine system and has an envelope conformation.
Download citation


Download citation


The product of the reaction of 3-ethoxycarbonylmethyl-2-oxo-1,2-dihydroquinoxaline with bromide was dissolved in dimethylformamide for further reaction with sodium azide in water at low temperature. The unexpected compound obtained has been structurally characterized. There is a short intramolecular contact between the N1 atom and a hydroxyl group [2.772 (4) Å]. Two intermolecular hydrogen bonds participate in the molecular packing.
Download citation


Download citation


The structure of lophine peroxide was confirmed to be 2,4,5-triphenyl-4H-imidazol-4-yl hydroperoxide and not the 2,5-endoperoxide. The asymmetric unit is composed of an O—H⋯N hydrogen-bonded dimer of lophine peroxide and a CH2Cl2 solvent molecule.
Download citation


Download citation


The structure of the title compound (a potential auxiliary in asymmetric alkylation) comprises cations and anions, linked by Cl⋯H—N—H⋯Cl hydrogen bonds (in which the F atom does not participate) into an infinite chain. The absolute configuration (S) was confirmed. The five-membered heterocycle adopts an envelope conformation, with the fluorodiphenylmethyl substituent in an equatorial orientation.
Download citation


Download citation


Hydrogen bonding in the three title compounds, (I>), (II>) and (III>), respectively, occurs between primary and quaternary amino, carboxylic acid and carbonyl groups of the dipeptides and the water solvate molecules. The tryptophan ring portions pack in a herring-bone sheet fashion, with no significant hydrogen-bonding interactions.
Download citation


Download citation


The title compound shows that the oxidative cyclization of 5-ethyl-3-(4-methoxybenzylidene)hydrazino-1,2,4-triazino[5,6-b]indole with ethanolic iron(II) chloride leads to the formation of linearly annelated species. The skeleton is approximately planar, except for the ethyl group.
Download citation


Download citation


The title compound is an inhibitor of neuronal nitric oxide synthase and is also a reversible monoamine oxidase B inhibitor. The planar benzenoid N1H tautomer crystallizes as a dimer assisted by intramolecular N—H⋯N hydrogen bonding.
Download citation


Download citation


The structure of the title compound shows that the cation and the bromide anion are linked via the water molecule through weak intermolecular hydrogen bonds.
Download citation


Download citation


The title compound, obtained as a minor component following acetylation and chromatographic fractionation of the mother liquor remaining after crystallizing castanospermine from extracts of Castanospermine australe seeds, is similar to previously determined australines, but with no intermolecular hydrogen bonding in the crystal.
Download citation


Download citation


The two cyclohexene rings adopt sofa conformations and are attracted towards each other by O—H⋯O intramolecular hydrogen bonds. C—H⋯O interactions connect adjacent molecules.
Download citation


Download citation


The title molecule is substantially planar. The phenyl and 3-chloro-4-fluorophenyl rings are on opposite sides of the C=N bond. There is an intermolecular C—H⋯F short contact and the molecules are held together in layers parallel to the bc plane.
Download citation


Download citation


The relative configuration was determined for the title compound, which was obtained as a mixture of epimers. There are both intra- and intermolecular hydrogen bonds, the latter forming dimers around the crystallographic centres of inversion.
Download citation


Download citation


Analysis of the title bis-(−)-menthylcarbonate derivative of trans-1,2-cyclopentanediylbis(2-benzenethiol), (II), determines the (−)-enantiomer of (II) to be S,S and the (+)-enantiomer to be R,R.
Download citation


Download citation


In the title compound, the amide groups are rotated out of the C(ONH2)—C(HPh)—C(ONH2) plane by ca 25–47° and the phenyl ring is almost perpendicular to this plane. Intermolecular N—H⋯O hydrogen bonds form a three-dimensional network.


 journal menu
journal menu