issue contents
April 2008 issue
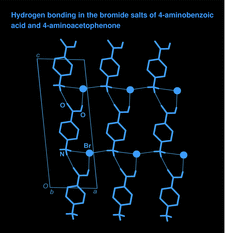
Cover illustration: A view of the two-dimensional hydrogen-bonded network of 4-carboxyanilinium bromide parallel to the (010) plane, showing the aggregation of three hydrogen-bonding motifs. Hydrogen bonds are drawn as thin lines and C-bound H atoms have been omitted. See Cincic & Kaitner [Acta Cryst. (2008), C64, o226-o229].
inorganic compounds
Download citation


Download citation


The Cu6P(Se0.7S0.3)5Br cubic structure is built from a [P(Se0.7S0.3)5Br] framework in which CuI ions are distributed in various tetrahedral, triangular and linear sites. The compound is a solid solution of two efficient ionic conductors, namely Cu6PSe5Br and Cu6PS5Br, in which high ionic conductivity results from order–disorder phenomena in the copper substructure.
Download citation


Download citation


The crystal structure of K2[O(HgSO3)3] features a layered assembly of K+ cations and complex [O(HgSO3)3]2− anions with m symmetry and a slightly distorted trigonal–pyramidal OHg3 core.
Download citation


Download citation


The title compound contains a unique framework of LiO4 and VO4 tetrahedra, which are linked to produce porous double layers separated along the c direction by isolated chains of face-sharing LiCl6 octahedra. Within the layers, six-tetrahedron Li2V4O18 rings form large channels filled by Ba2+ and Cl− ions.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu