issue contents
June 2012 issue
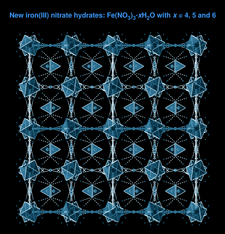
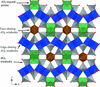
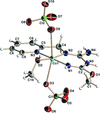
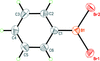
Cover illustration: 2 × 2 × 2 unit cells of Fe(NO3)3·6H2O, viewed in the a-axis direction. See Schmidt, Asztalos, Bok & Voigt [Acta Cryst. (2012), C68, i29-i33].
inorganic compounds
Download citation


Download citation


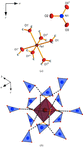
The crystal structure of iron(III) trinitrate hexahydrate is built up from [Fe(H2O)6]2+ octahedra and nitrate anions connected via hydrogen bonds. In iron(III) trinitrate pentahydrate, one water molecule in the coordination octahedron of the FeIII atom is substituted by an O atom of a nitrate group. Iron(III) trinitrate tetrahydrate represents the first example of a simple iron(III) nitrate with pentagonal–bipyramidal coordination geometry, where two bidentate nitrate anions and one water molecule form a pentagonal plane.
Download citation


Download citation


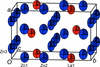
The compound Li3Al(MoO4)3 adopts the lyonsite crystal structure type. The edge-sharing and face-sharing octahedral sites exhibit disorder between Li+ and Al3+.
Download citation


Download citation


The structure of LaZn4 has been determined from single-crystal X-ray diffraction data for the first time, approximately 70 years after its first discovery. The structure is closely related to the BaAl4, La3Al11, BaNi2Si2 and CaCu5 structure types, which can be presented as close-packed arrangements of 18-vertex clusters, in this case LaZn18.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu