issue contents
January 2016 issue
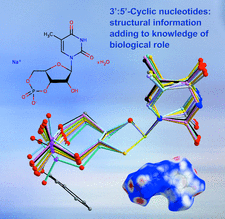
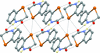
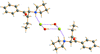
Cover illustration: Two salts of thymidine 3′:5′-cyclic phosphate (cdTMP), namely Na(cdTMP)·7H2O, (I), with Z′ = 1, and Na(cdTMP)·3.7H2O, (II), with Z′ = 8, are compared in terms of cyclic nucleotide conformation, inter-nucleotide interactions and nucleotide–sodium interactions. See Ślepokura [Acta Cryst. (2016), C72, 35–47].
research papers
Download citation


Download citation


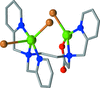
The structures of a six- and a seven-membered tellurium- and nitrogen-containing (Te—N) heterocycle represent the first structurally characterized members of their respective classes. When compared to their sulfur analogs, they exhibit slightly greater deviations from planarity to accommodate the larger chalcogenide atom. Intermolecular N—H⋯O hydrogen bonding forms centrosymmetric ![[R_{2}^{2}]](/c/issues/2016/01/00/sk3604/teximages/sk3604fi1.gif) (8) dimers in the six-membered hetereocyclic compound and chains in the seven-membered hetereocyclic compound, while intermolecular Te⋯O contacts exist in the former.
(8) dimers in the six-membered hetereocyclic compound and chains in the seven-membered hetereocyclic compound, while intermolecular Te⋯O contacts exist in the former.
![[R_{2}^{2}]](/c/issues/2016/01/00/sk3604/teximages/sk3604fi1.gif) (8) dimers in the six-membered hetereocyclic compound and chains in the seven-membered hetereocyclic compound, while intermolecular Te⋯O contacts exist in the former.
(8) dimers in the six-membered hetereocyclic compound and chains in the seven-membered hetereocyclic compound, while intermolecular Te⋯O contacts exist in the former. Download citation


Download citation


Changes in the heating rate and reaction temperature leads to the formation of four supramolecular isomers of dichloridobis(1,10-phenanthroline)cobalt(II) from dimethylformamide. The structures and isomerization behaviours of the four isomers are discussed.
Download citation


Download citation


Two building blocks having a major influence on the stereochemical orchestration of the drug molecule eribulin mesylate were prepared and their two-dimensional nuclear magnetic resonance (2D-NMR) spectra generated. These data were corroborated by the single-crystal X-ray diffraction data generated for these molecules.
Download citation


Download citation


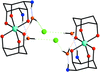
A two-dimensional cyanide-bridged bimetallic coordination polymer [CuIICoIII(CN)6(Hpip)(H2O)]n (Hpip is piperazinium) exhibits a two-dimensional (4,4) grid-like structure, in which cationic [CuII(Hpip)(H2O)]3+ and anionic [CoIII(CN)6]3− units are linked through bridging cyanide ligands. The grid is further extended into a three-dimensional supramolecular network through hydrogen-bonding interactions. A magnetic investigation shows antiferromagnetic coupling in the compound.
CCDC reference: 1405770
Download citation


Download citation


In [Ge(taci)2]2Cl8·12H2O (taci is 1,3,5-triamino-1,3,5-trideoxy-cis-inositol), the GeIV centre is exclusively bonded to the O-atom donors of two taci moieties. The taci ligand adopts a zwitterionic form with deprotonated hydroxy groups and protonated amino groups. Extended hydrogen bonding constitutes a complex three-dimensional network.
CCDC reference: 1440044
Download citation


Download citation


Two salts of thymidine 3′:5′-cyclic phosphate (cdTMP), namely Na(cdTMP)·7H2O, (I), with Z′ = 1, and Na(cdTMP)·3.7H2O, (II), with Z′ = 8, are compared in terms of cyclic nucleotide conformation, inter-nucleotide interactions and nucleotide–sodium interactions.
Download citation


Download citation


The dangerous new drug α-PVP crystallizes as the chloride salt with slightly less than a full solvent water molecule, forming hydrogen bonds between each of the water H atoms and the chloride ion, which is itself hydrogen bonded to the quaternary N atom of the drug cation. A comparison with the structure of a number of other bath salts shows that, even though the bond between the phenyl group and the ketone group has free rotation, the conformation is essentially always the same.
CCDC reference: 1411976
Download citation


Download citation


Two related fused tetracyclic benzo[b]pyrimido[5,4-f]azepine derivatives exhibit very different conformations for the azepine ring and very different patterns of supramolecular assembly.
Download citation


Download citation


meso-(E,E)-1,1′-[1,2-Bis(4-chlorophenyl)ethane-1,2-diyl]bis(phenyldiazene) was formed in high yield in the reaction between 4-chlorobenzaldehyde and 1-phenylpyrazolidine-3,5-dione, and crystallized from ethanol to give triclinic and monoclinic polymorphs, where the monoclinic polymorph exhibits configurational disorder. The only significant intermolecular contacts are of the C—Cl⋯π(arene) type in the triclinic polymorph and of the C—H⋯N and C—H⋯π(arene) types in the monoclinic polymorph.
Download citation


Download citation


A three-dimensional Cu+-containing coordination polymer, formed from the in situ reduction of CuCl2 in the presence of 3,5-diaminobenzoic acid and KOH, contains ten distinct CuI atoms, two of which lie on crystallographic inversion centres. Each CuI atom is associated with two adjacent metal atoms and if all these are regarded as `cuprophilic' interactions, then infinite [![[\overline{1}]](/c/issues/2016/01/00/sk3607/teximages/sk3607fi1.gif) 01] zigzag chains of CuI atoms occur in the crystal.
01] zigzag chains of CuI atoms occur in the crystal.
![[\overline{1}]](/c/issues/2016/01/00/sk3607/teximages/sk3607fi1.gif) 01] zigzag chains of CuI atoms occur in the crystal.
01] zigzag chains of CuI atoms occur in the crystal.CCDC reference: 990165
Download citation


Download citation


Cu2(tpena)Br3 and Zn2(tpena)X3 [X = Br or Cl and tpena = N,N,N′-tris(pyridin-2-ylmethyl)ethylenediamine-N′-acetate] show distinctly different molecular structures despite being indistinguishable using routine spectroscopic techniques. The ligand is unexpectedly dinucleating in the case of the copper complex, while it bridges to a [ZnX3]− unit in the zinc complexes.
Download citation


Download citation


The structure analyses of three benzoylhydrazine–dithiocarbazoate derivatives support the hypothesis that planarity of their molecules plays an important role in their tuberculostatic activity.
Download citation


Download citation


A new V4+ layer structure, V2O2F4(H2O)2·H2O, built up of VOF4(H2O) corner- and edge-sharing octahedral building units, with structural relationships to previously synthesized vanadium oxyfluoride compounds, has been synthesized via solvothermal methods.
CCDC reference: 1442717
Download citation


Download citation


6-Chloroisocytosine has been cocrystallized with two coformers in an approach intended to obtain the donor–donor–acceptor/acceptor–acceptor–donor synthon, as well as to determine the tautomerism.

 journal menu
journal menu