issue contents
November 1998 issue
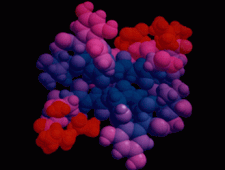
Cover illustration: The balhimycin dimer, with citrate and acetate ions in the binding pockets, colour coded on a scale where the most mobile atoms are red and the least mobile are blue. The binding pockets are clearly the most mobile, consistent with the idea that they can open and close to complex with cell-wall binding peptides. Courtesy of George Sheldrick.
research papers
Boronic acid-containing inhibitors of human α-thrombin were elaborated using structure-based design approaches. Introduction of substituents to an inhibitor aromatic ring modulated the strength of an edge-to-face interaction and, in other cases, resulted in a new inhibitor binding mode.
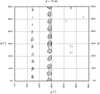
The variation in the extent of the orientational ordering of protein molecules has been revealed for tetragonal lysozyme single crystals of different quality on the basis of their polarized Raman spectra.
The joint probability distribution of electron density and its gradient in protein electron-density maps was found to be independent of structural conformation and sensitive to phase error. This suggests that it could be used as a constraint for protein phase refinement and extension.
The crystal structure of a designed variant of the ROP protein has been refined to a resolution of 1.09 Å. Modelling disorder and refinement of anisotropic motion played a signicant role in achieving convergence.
PDB reference: ColE1 ROP variant, 1nkd
In the structure determination of echovirus 1, subtle interactions between crystallographic and non-crystallographic symmetries caused the space group to be ambiguous, but also allowed significant short cuts in the molecular-replacement calculations.
PDB reference: echovirus1, 1ev1
A new Z-DNA structure d(TGCGCA)2 determined at 120 K to a resolution of 1.3 Å was compared with other DNA crystal structures. Their packing and interaction with cations and water is discussed.
PDB reference: d(TGCGCA)2, 362d
The power of maximum-likelihood structure refinement is increased further by the addition of prior phase information.
The first near-atomic resolution structure of a T = 4 virus was determined with progressively higher resolution models for phase calculation and 60-fold non-crystallographic symmetry for phase refinement and extension. Phases at low resolution were computed with a hollow sphere and they were refined and extended to higher resolution which allowed more detailed models to be fit to the density and used for phase computation for the next round of phase extension.
Average multipole parameters obtained from charge-density studies of peptides have been transferred to a 310 helix octapeptide structure at 0.82 Å resolution. The improvement of the crystal structure as well as the limits of application to oligopeptides and proteins of the multipole transfer are discussed.
Refinement of the structure of human apolactoferrin at 2.0 Å resolution has revealed a possible mechanism whereby conformational changes in the N-lobe of the protein can be signalled to the C-lobe.
The revived application of the classical molecular Fourier transform method has been used to solve the structures of proteins and DNA from X-ray diffraction data.
The structures of a new crystal form of ribonuclease A and its low-humidity variant have been determined. A comparison of these structures with those of other forms provide information on the plasticity of the protein molecule, role of invariant water molecules and the relation between accessibility and hydration.
The crystal structure of spinach ferredoxin I provides a structural framework for the interpretation of the large amount of data on this protein present in the literature.
PDB reference: spinach ferredoxin I, 1a70
In order to assess the feasibility of time-resolved studies on the enzyme acetylcholinesterase, a Laue data set with a total exposure time of 24 ms has been collected. Electron-density maps clearly show the soaked-in ligand as well as a small structural change in the active site.
The work describes the first crystal structure of the human papillomavirus serotype 31 E2 DNA-binding domain. The overall structure is discussed, novel features are described, and implications to be previous work are given.
PDB reference: human papillomavirus serotype 31 E2 DNA-binding domain, 1a7g
HREM observation of octocalcium phosphate (OCP) in human dentine crystal and of its hydrolysis to hydroxyapatite is described. OCP is a transient phase during biological crystal growth.
The crystallization of T. thermophilus tRNAAsp complexed with its cognate aspartyl-tRNA synthetase is described. The Matthews coefficient has been determined and compared with those obtained for other aminoacylation systems.
Strategies to produce protein crystals benefit tremendously from knowledge of the protein's solubility and the rate of crystal growth as a function of temperature and initial sample composition. This article describes a method to obtain such information within a matter of a few days utilizing a modest amount of protein in a nondestructive fashion via isothermal microcalorimetry.
crystallization papers
Crystals of archaeal O6-methylguanine–DNA methyltransferase from Pyrococcus kodakaraensis strain KOD1 have been grown and diffract to 2.0 Å resolution upon exposure to Cu Kα X-rays.
Crystals of the pyridoxal-phosphate-dependent aminotransferase involved in the antepenultimate step in biotin biosynthesis have been obtained and analyzed with respect to cell dimensions, space group and suitability for a crystallographic structure determination.
OmpC osmoporin from E. coli has been crystallized under a variety of conditions. The crystals belong to space group P21 and diffract to 4 Å.
Diffraction-quality crystals of the unliganded mouse recombinant catalytic subunit of cAMP-dependent protein kinase have been grown by the hanging-drop vapour-diffusion technique using 2-methyl-2,4-pentanediol as precipitant and preliminary X-ray analysis has been carried out.
Tropinone reductase II, an NADPH-dependent oxidoreductase involved in a plant alkaloid metabolism, has been crystallized using a macroseeding technique. A data set to 2.6 Å resolution has been collected at a cryogenic temperature.
Crystals of a complex between ovine placental lactogen, a close prolactin homologue, and two copies of the extracellular portion of the rat prolactin receptor have been grown. The crystal structure of this complex will shed light on the structural reasons for cross-reactivity and specificity among the endocrine hormones, placental lactogen and growth hormone.
The chitinase from C. immitis has been crystallized in a form suitable for high-resolution diffraction studies.
Prolyl oligopeptidase, a member of a new serine protease class, has been crystallized in the presence of an inhibitor. The crystals diffract to 2.3 Å resolution.
Crystals of the R. meliloti DctD two-component receiver domain, expressed in E. coli and purified to homogeneity, were obtained using the hanging-drop vapor-diffusion method. They diffract to 2.3 Å and exhibit the symmetry of space group I222 or I212121 with cell dimensions a = 59.0, b = 58.6, and c = 169.8 Å.
The triosephosphate isomerase (TIM) from the Archaeon hyperthermophilic is tetrameric, in contrast to most other TIMs, which are dimeric. The crystallization of this hyperthermophilic TIM is reported and should lead to an understanding of the nature of protein thermostability in the enzyme.
Well ordered crystals of the enzyme 6-phosphogluconate dehydrogenase from L. lactis have been obtained.
The crystallization of C. perfringens α-toxin (from two different strains), the key determinant of gas-gangrene disease, is reported, in two different forms. The structure of this toxin will provide important new information towards elucidating the pathogenesis of this often lethal disease.
The flavoenzyme polyamine oxidase plays an important role in the regulation of polyamine intracellular concentration. Crystals of maize polyamine oxidase suitable for structure determination have been grown using ammonium sulfate as precipitant.
Crystallization and preliminary X-ray diffraction study of lactonohydrolase from Fusarium oxysporum
The lactonohydrolase from F. oxysporum was crystallized in the presence of polyethylene glycol 4000 as a precipitant. The crystals belong to the monoclinic space group P21 with unit-cell parameters a = 156, b = 100, c = 94.1 Å, β = 91.7°.
The homing endonuclease I-DmoI has been crystallized. Crystals diffract to 2.3 Å resolution and are suitable for structure determination.
Piratoxin II, a phospholipase A2 from B. pirajai has been crystallized in a monoclinic space group and its structure has been solved by molecular replacement at 2.04 Å resolution.
The crystallization and preliminary X-ray analysis of T. serrulatus neurotoxin Ts-γ is reported. It crystallizes in space group P21 with one molecule per asymetric unit has been solved by molecular replacement at 1.73 Å resolution.
Analysis of the X-ray diffraction data collected on single crystals of a calcium-binding protein from E. histolytica suggests that the protein resembles the canonical calmodulin.
The seed lectin DBL and the related stem and leaves lectin DB58 of the tropical legume Dolichos biflorus were crystallized, as well as complexes of DBL with adenine and with GalNAc(α1-3)[Fuc(α1-2)]Gal.
Human plasma apolipoprotein H (Mw ≃ 50 kDa) plays an important role in blood coaguilation and antiphospholipid autoimmune disease. With synchrotron radiation, apolipoprotein H crystals diffracted to 3.0 Å resolution and belong to the orthorhombic space group C2221 with a = 158.47, b = 169.25, c = 113.28 Å (at 100 K).
Crystals of the DNA-binding protein GerE from B. subtilis have been grown in a form suitable for X-ray diffraction studies.
Crystals of an antibody fragment specific to the blood-group A carbohydrate antigen have been grown and diffract to 0.93 Å resolution. The protein was expressed from a synthetic gene construct, and the kinetics of antibody–antigen binding have been determined by surface plasmon resonance.
The PhoB receiver domain has been crystallized and X-ray diffraction data to 1.88 Å resolution have been collected using synchrotron radiation. The crystals contain two molecules per asymmetric unit, suggesting that the active dimer has been formed.
short communications
The pneumococcal surface antigen PsaA has been produced and crystallized. The structure of this protein is currently being determined via multiple isomorphous replacement.
We have crystallized yeast glyceraldehyde 3-phosphate dehydrogenase in order to use this enzyme as a probe for possible enzyme–enzyme interactions.
fast communications
The N-terminal transactivation domain of the E2 protein from human papillomavirus type 16 has been crystallized. This domain interacts with both the HPV E1 protein and at least three cellular transcription factors.
international union of crystallography
Free 



 journal menu
journal menu