issue contents
April 1999 issue
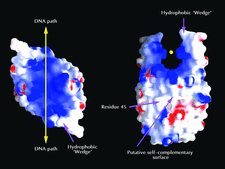
Cover illustration: The surface electrostatic potential of protein HU from B. stearothermophilus showing the putative protein-DNA and protein-protein interaction sites (p. 801).
scientific comment
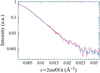
A low-resolution structure from X-ray scattering data of Kp1·(ADP·AlF-4·Kp2)2 predicted a significant change in the iron protein (Kp2) upon complex formation. This has been subsequently confirmed by the crystallographic structure of the complex in the Av system. New scattering results are provided to demonstrate the similarity of this complex in the two species.
research papers
The terminally brominated DNA octamer duplex d[ACGTACG(5-BrU)]2 has been used in an ab initio structure determination in a four-wavelength MAD experiment, and the result used in a comparison of the effect of replacing Me by Br on the DNA duplex using our previous results on the native analogue d(ACGTACGT)2.
NDB reference: d[ACGTACG(5-BrU)]2, adhB99
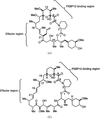
The X-ray structure of the FKBP12–rapamycin–FRB complex refined at 2.2 Å clarifies how the immunosuppressive drug rapamycin simultaneously binds two different proteins, FKBP12 and FRB.
PDB reference: FKBP12–rapamycin–FRB ternary complex, 2fap
A small improvement in the bound-solvent structure is seen in lysozyme crystals grown in microgravity by liquid–liquid dialysis, which has a more stable fluid physics state, and is consistent with a better formed protein crystal in microgravity.
The structure of an orthorhombic crystal form of α2u-globulin (the major urinary protein excreted by adult male rats) and that of a complex of this protein with d-limonene 1,2-epoxide, a compound which causes renal carcinogenesis in male rats, is described. The specific make-up of the ligand-binding cavity provides insight into the structural basis of the specificity of the renal carcinogenesis for male rats.
The orientation and position of the T = 7 icosahedral capsid of the dsDNA phage HK97 were determined at 7 Å resolution. The particle is roughly 600 Å in diameter and is the first dsDNA phage capsid studied by X-ray crystallography.
The X-ray crystal structure of mouse tumour-necrosis factor has been determined to 1.4 Å and is compared with human tumour-necrosis factor structures. Some loop regions important for receptor binding are different and may explain the species specificity of receptor binding.
PDB reference: mouse tumour-necrosis factor, 2tnf
Determination of the structure of Pp acetylxylan esterase by an iodination method is described. The structures of the native protein and the iodinated complex are presented at 1.10 and 1.80 Å, respectively.
The selectide inhibitiors bind to thrombin in a retro fashion making a parallel β-strand with Ser214–Gly216. Since they are 104 times more specific for factor Xa, if factor Xa binding is also retro, the selectivity can be a consequence of S3–S4 subsite binding interactions.
The structure of the basic lectin from winged bean has been determined and compared with the structure of its complex with methyl-α-D-galactoside.
PDB reference: saccharide-free basic form of winged-bean lectin, 1wbf
The structure of DNA-binding protein HU has been determined and refined at 2.0 Å resolution. The structural basis of HU's ability to bind, bend and supercoil DNA is discussed.
PDB reference: DNA-binding protein HU, 1huu
The packing arrangement of oligonucleotides crystallized in the B form is reviewed. It is shown that packing interactions have a strong influence on the average conformational parameters of DNA.
A known small protein, rubredoxin, which contains 52 amino-acid residues including an FeS4 unit, a sulfate ion and 102 solvent water molecules has been solved ab initio by the direct-methods program SAYTAN. The model is refined to R = 14.5% using 1 Å resolution data.
Difficulties encountered in searching for two tetramers of human glucose 6-phosphate dehydrogenase (Canton variant) using a bacterial G6PD dimer as model were resolved by finding a tetramer in a simpler crystal form and using a tetramer search model. The human G6PD subunit was a suitable model to solve a deletion mutant with crystallographic 222 symmetry.
Following phase determination by partial-model molecular replacement, weak single isomorphous replacement and inter-crystal averaging, conventional refinement led only to overfitting and not to model improvement. Progress was dependent on modest improvements achieved without overfitting using refinement by a local real-space method.
A multisolution direct method has been proposed to resolve the phase ambiguity intrinsic in single isomorphous replacement data of proteins with the replacing atoms in a centrosymmetric arrangement. A test with experimental data produced satisfactory results.
Open  access
access
 access
accessA fully automated procedure for solving MIR and MAD structures has been developed using a scoring scheme to convert the structure-solution process into an optimization problem.
crystallization papers
Two different crystal forms of the complex of bovine factor Xa and recombinant tick anticoagulant peptide have been obtained using PEG as precipitant.
Glutamine synthetase from M. tuberculosis has been crystallized in an orthorhombic space group with two dodecamers (total molecular weight of 1.3 MDa) in the asymmetric unit. Data to 2.4 Å resolution yield the crystal packing from self-rotation calculations, Matthews number, and a native Patterson map.
Two crystal forms of α-glucuronidase from the thermophilic bacterium B. stearothermophilus T-6 have been obtained recently, both of which diffract to better than 3 Å resolution and are suitable for a full crystal structure analysis.
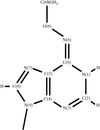
The crystal of the DNA dodecamer of d(CGCGmo6AATCCGCG) containing 2′-deoxy-N6-methoxyadenosine changes from one form to another during data collection, as a phase transition ascribed to differences in humidity. It has been found as the first example that the N6-methoxyadenosine residue forms a base pair with cytosine residue in a manner identical to Watson–Crick-type pairing.
Periplasmic nitrate reductase from D. desulfuricans ATCC 27774 has been crystallized by the vapour-diffusion method using polyethylene glycol as precipitant. Data collection under cryogenic conditions allowed the determination of the space group (P3121 or P3221) and the unit-cell parameters (a = b = 106.3, c = 135.1 Å).
A report is presented of the expression and crystallization of the horse lipocalin Equ c 1, the major component responsible for the induction of specific IgE antibodies in patients sensitized to horse allergens.
Two orthorhombic forms of crystals of the haptoglobin–hemoglobin complex are described. They did not diffract well enough for data collection.
Crystals have been characterized of an integral membrane light-harvesting complex with different spectral characteristics to other LH2 complexes. The elucidation of this structure will allow the role that the protein plays in modulating the absorption properties of the bacteriochlorophyll a pigments to be determined.
The human spliceosomal U5 snRNP-specific 15 kDa protein as well as a selenomethionine derivative of the protein were overproduced in Escherichia coli, purified and crystallized. Well diffracting single crystals were obtained.
The membrane-bound oxygen-evolving reaction centre complex of photosystem II from both spinach and pea (Mr ≃ 250 kDa) have been crystallized in a variety of mixtures of detergents. The crystals diffracted to 6.5 Å resolution.
A truncated form of the β subunit of human protein kinase CK2 has been overproduced and crystallized allowing its X-ray structure determination.
A truncated β-amylase from B. polymyxa has been crystallized. The crystals contain three protein molecules in the asymmetric unit and diffract to 2.5 Å resolution.
The Fe(III)-dependent intradiol dioxygenase hydroxyquinol 1,2-dioxygenase from N. simplex, has been crystallized in space group P212121. A native data set, complete to 1.8 Å, has been collected. The search for suitable heavy-atom derivatives is in progress.
Crystallization of hydroxynitrile lyase from M. esculenta in two different space groups and X-ray diffraction analysis are presented.
A highly diffracting crystal of K. oxytoca diol dehydratase complexed with cyanocobalamin has been produced and preliminary crystallographic data obtained.
An electron-density map of an adequate quality to allow model-building of the M. tuberculosis chaperonin 10 structure has been obtained by using the non-crystallographic symmetry relating the subunits in phase extension from an initial molecular-replacement model. Analysis of the phase-extended 2Fo − Fc map reveals two heptamers interact through conformationally flexible loops.
A novel bacterial esterase has been crystallized in two crystal forms allowing high-resolution data to be collected.
A His-tagged form of the κ-carrageenase from P. carrageenovora has been overproduced and crystallized allowing its X-ray structure determination.
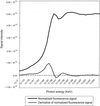
The selenomethionine-substituted catalytic subunit of the E. coli arsenite transporter has been crystallized. Initial studies have identified the positions of 11 out of 13 methionines and indicate that structure determination by MAD phasing is feasible.
The novel phospholipase A2 from D. russelli pulchella has been crystallized using the hanging-drop vapour-diffusion method with ammonium sulfate as precipitating agent. Crystals belong to the orthorhombic space group C2221 with unit-cell dimensions a = 77.01, b = 92.29, c = 76.90 Å with two molecules in the asymmetric unit.
Glutamate racemase from A. pyrophilus has been purified and crystallized by the hanging-drop vapor-diffusion method using polyethylene glycol 6000 as a precipitant. The crystals belong to space group P6122 or P6522 with unit-cell dimensions of a = b = 72.1 and c = 185.02 Å.
The crystallization conditions of β-1,4-galactanase from A. aculeatus have been optimized. The crystals have been characterized by X-ray diffraction and diffract to 2.3 Å. This is the first enzyme of family 53 of the glycosyl hydrolases to be crystallized.
Maltooligosyl trehalose synthase has been crystallized and preliminary X-ray diffraction data have been obtained. The chemical modification of the enzyme improved crystal quality.
short communications
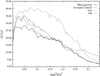
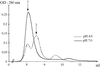
The structures of orthorhombic lysozyme grown at basic pH and its low-humidity variant have been determined. Comparison with structures of crystals grown at acidic pH does not indicate any systematic pH-dependent variations in molecular geometry. The changes in the molecule during the transformation to the low-humidity form are more pronounced in the C-terminal region.
An enhanced version of MolScript is described. Amongst its new features, the ability to represent electron-density maps is of particular interest to crystallographers.
Software tools for generating, optimizing, handling, storing and displaying macromolecular envelopes (masks) are described.


 journal menu
journal menu