issue contents
June 1999 issue
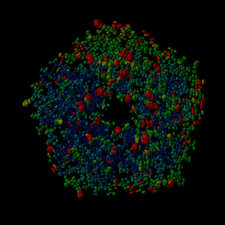
Cover illustration: Thermal ellipsoid representation, generated by Raster3D, of atomic anisotropy in the structure of the cholera toxin B-pentamer refined at 1.25 Å (p. 1109).
topical reviews
The distribution of atomic anisotropy is proposed as a validation tool for protein structures refined with individual anisotropic displacement parameters Uij. This distribution is examined for all such protein models currently in the Protein Data Bank.
research papers
Atomic resolution refinement of the Y43F mutant of streptavidin shows 2-methyl-2,4-pentanediol molecules in the biotin binding site.
PDB reference: Y43F streptavidin mutant, 1swu
The crystal structures of two isoforms of nucleoside diphosphate kinase from bovine retina with nucleoside in the active site are reported to 2.4 Å resolution.
The crystal structure contains two A-DNA decamer duplexes of d(G4CGC4) in the asymmetric unit. The structural differences between the two molecules appear to originate from an inherent propensity of the sequence for varied intermolecular interactions and malleability.
NDB reference: d(G4CGC4), ad0002
The secondary and tertiary structure of β-momorcharin is similar to other single-chained ribosome-inactivating proteins (RIPs). The oligosaccharide is linked to the protein through an N-glycosidic bond, stretches from the molecular surface far from the active site and interacts with the protein through hydrogen bonds, most of them being intermolecular interactions with the protein atoms in an adjacent unit cell.
PDB reference: β-momorcharin, 1cf5
The structure of mare apolactoferrin shows that both N and C lobes adopt closed conformations. Thus, the structure of mare apolactoferrin is different from the structures of human apolactoferrin and duck apoovotransferrin.
PDB reference: mare apolactoferrin, 1b7u
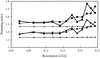
SHELX refinement strategies at medium resolution are illustrated for an immunoglobulin fragment. The effect of local NCS restraints, anisotropic scaling and anisoropic refinement has been tested, and standard uncertainties calculated.
PDB reference: RELv-T39K, 1bww
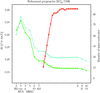
A multiwavelength anomalous diffraction experiment has been completed in 45 min using synchrotron radiation from the Structural Biology Center's 19ID undulator beamline at the Advanced Photon Source.
PDB reference: chaperonin apical domain, 1srv
Open  access
access
 access
accessA reciprocal-space measure of the quality of macromolecular crystallographic phases based on the variance of the local roughness of the map is presented.
An improvement for a statistical test aimed at detecting twinning and a new method to determine the level of twinning, as well as a new parametrization for twinning, are proposed.
Low-salt crystallization of T7 RNA polymerase: a first step towards the transcription bubble complex
New low-salt crystallization conditions have been obtained for T7 RNA polymerase and have been used to produce crystals of the native enzyme, a chimeric T7/T3 RNA polymerase and native T7 RNA polymerase containing an N-terminal histidine tag.
crystallization papers
The crystals of acuthrombin-B, a thrombin-like enzyme consisting of two non-identical polypeptide chains (14.4 and 16 kDa) from A. acutus venom, bleong to space group P21 with a resolution limit near 2 Å and unit-cell parameters a = 34.97, b = 53.58, c = 67.88 Å, β = 98.89°, and contain one molecule per asymmetric unit.
Crystallization and preliminary X-ray diffraction analysis of a biologically active fragment of CD55
Crystals have been grown of a virus-binding construct of CD55 which diffract X-rays to at least 1.65 Å.
Ferrochelatase of D. melanogaster has been purified and crystallized with an intact [2Fe–25] cluster. The crystals diffract to 3.0 Å resolution with a space group of I422.
Preliminary X-ray characterization of the two orthorhombic and one hexagonal crystal forms of a basic lectin from A. hirsuta is reported.
The formiminotransferase domain of formiminotransferase cyclodeaminase, a bifunctional channeling enzyme involved in folate metabolism, has been crystallized in space group P212121. Native diffraction data and data for a mercurial derivative have been collected to 2.8 Å resolution.
Cytochrome P450sca-2 from S. carbophilus has been crystallized using ammonium sulfate as precipitant. The crystals diffract to 2.2 Å resolution.
Crystallization and preliminary X-ray analysis of the 6-phospho-α-glucosidase from Bacillus subtilis
Crystals of GlvA, the 6-phospho-α-glucosidase from B. subtilis, have been grown in a form suitable for X-ray diffraction studies.
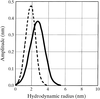
A novel purification method of the oxygen-sensor domain of R. meliloti FixL, dynamic light-scattering studies of its solution and preliminary crystallographic data are reported.
Recombinant S. cerevisiae Ypd1p fused with a His6-tag at its C-terminus has been crystallized. The triclinic crystal contains four monomeric Ypd1p in the asymmetric unit and native data have been collected to 2.3 Å.
A periplasmic tetrahaem flavocytochrome c3 from S. frigidimarina NCIMB400 which possesses fumarate reductase activity has been crystallized. The crystals belong to space group P212121 with unit-cell parameters a = 72.4, b = 110.1, c = 230.2 Å. Assuming a molecular dimer in the asymmetric unit, the crystals contain 65% solvent and, when cryocooled to 100 K, diffract to at least 3.0 Å resolution. An iron X-ray absorption edge has been measured in preparation for a MAD phasing experiment.
Recombinant phosphopantetheine adenylyltransferase, a key late enzyme in coenzyme A (CoA) biosynthesis from E. coli has been crystallized in complex with various substrates, its inhibitor (CoA) and as the apo form.
Piratoxin III, a D-49 phospholipase A2 from B. pirajai has been crystallized in the monoclinic space group C2 and its structure has been solved by molecular replacement at 2.7 Å.
Hydrolytic haloalkane dehalogenase from S. paucimobilis UT26 has been crystallized by the vapour-diffusion method using PEG 6000 as a precipitant. Native diffraction data were collected to 1.6 Å resolution under cryogenic conditions.
S. cerevisiae Hsp40 Sis1 has been purified and crystallized. The crystals diffract to 2.7 Å and belong to space group P41212 or P43212.
Hn-33, a protein in the botulinum neurotoxin type A toxic complex, has been crystallized using vapour-diffusion techniques. The crystals belong to an orthorhombic space group and diffract to a resolution of 2.6 Å.
A novel complex of phospholipase A2 complexed with another venom protein has been isolated and purified from saw-scaled viper (E. carinatus) venom. It was crystallized with 7%(w/v) PEG 4000 and the crystals belonged to monoclinic space group P21 with unit-cell parameters a = 74.47, b = 47.87, c = 106.19 Å.
Cadmium-induced metallothionein II from rabbit liver has been crystallized in space group P6222 or P6422. Unit-cell parameters have been obtained for crystallization from sodium citrate and Tris–HCl buffers.
A 1:2 complex between the Bowman–Birk trypsin inhibitor from barley seeds and porcine pancreatic trypsin has been crystallized. Native X-ray diffraction data have been collected to 2.2 Å.
short communications
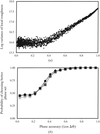
Fractal surfaces are effective and promising in the crystallization of macromolecules using the sitting-drop vapour-diffusion method.


 journal menu
journal menu