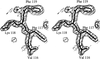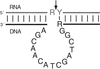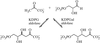issue contents
November 1999 issue
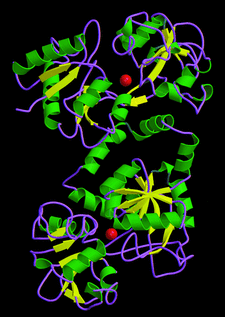
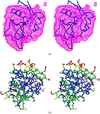
Cover illustration: Three-dimensional folding of buffalo lactoferrin (p. 1805).
research papers
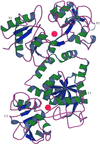
Ab initio solution and refinement of two high-potential iron protein structures at atomic resolution
Structures of a new crystal modification of the high-potential iron protein (HiPIP) from C. vinosum and its H42Q mutant have been solved by direct methods and refined anisotropically against data to 1.20 and 0.93 Å, respectively.
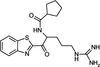
The crystal structure of covalently complexed inhibitor RWJ-51084 with bovine pancreatic β-trypsin at 1.8 Å resolution explains the specificity of the inhibitor for trypsin.
PDB reference: RWJ-51084–bovine pancreatic β-trypsin complex, 1qcp
The structure of oxalate-substituted lactoferrin has been determined at 2.7 Å resolution. The structure shows that the substitution of an oxalate anion does not perturb the overall structure of the protein but produces several significant changes at the metal and anion binding sites.
PDB reference: oxalate-substituted diferric mare lactoferrin, 1b7z
The structure analysis of a complex of lactoferrin with a lanthanide ion (Sm3+) shows that the protein is capable of sequestering ions of different sizes and charges, though with reduced affinity.
PDB reference: lactoferrin–Sm3+ complex, 1qjm
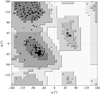
The inter-lobe orientations in buffalo lactoferrin at 277 and 303 K are shown to be different by 9°. The domain orientations at these two temperatures are identical.
PDB reference: buffalo lactoferrin, 1ce2
The cryo-cooled 90 K structure of a double-headed α-chymotrypsin inhibitor from winged bean seeds at 2.13 Å resolution exhibits conformational changes in the loops and side chains of some surface residues while retaining the overall conformation observed in the room-temperature structure.
PDB reference: winged bean chymotrypsin inhibitor, 4wbc
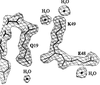
The structure of phosphoglycerate mutase:3-phosphoglyceric acid complex has been determined showing 3-phosphoglyceric acid binding at the base of a 12 Å cleft.
The structure of T. thermophilus L30 is presented to 1.9 Å resolution and compared with L30 from B. stearothermophilus.
PDB reference: ribosomal protein L30, 1bxy
The structure of acutolysin-C is reported, providing further evidence for the mechanism of the pH-dependent proteolytic reaction of zinc metalloproteinases.
PDB reference: acutolysin-C, 1qua
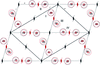
The crystal structure of the histidine-containing phosphotransfer (HPt) domain of the anaerobic sensor kinase ArcB from E. coli was refined to 1.57 Å resolution, using the coordinates of the earlier 2.06 Å structure as a starting model.
PDB reference: HPt domain of ArcB, 2a0b
The structures of three complexes of cellular retinoic acid binding proteins I and II with various synthetic retinoid compounds are described. The structures provide insight into the flexibility of these proteins in accommodating structurally diverse retinoid ligands.
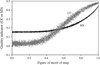
A phase transition has been observed in monoclinic factor XIII crystals upon transferring them to mother liquor containing high concentrations of the precipitant PEG 6000. The transition has been analyzed by X-ray crystallography and most of the change can be explained by small movements of the factor XIII dimers in the crystal lattice.
Open  access
access
 access
accessA procedure is described for improvement of crystallographic phases by reciprocal-space maximization of a likelihood function including experimental phases and characteristics of the electron-density map.
Open  access
access
 access
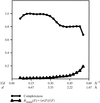
accessThe correlation of local r.m.s. density is shown to be a good measure of the presence of distinct solvent and macromolecule regions in macromolecular electron-density maps.
A method is described that allows for assessment and validation of differences between related protein crystal structures using experimental crystallographic information alone.
A procedure has been developed for identifying the optimal sizes of a multi-stranded nucleic acid complex that provide diffraction-quality crystals.
The joint probability distribution of the electron density and its gradient was used as a constraint in a two-dimensional histogram-matching procedure for map and phase improvement. The two-dimensional histogram-matching method was shown to be more powerful in phase refinement and extension than the one-dimensional histogram-matching method.
crystallization papers
Nitroreductase from E. coli has been crystallized in the space group P1. Diffraction data have been collected to 2.3 Å using cryocooling methods.
Preliminary X-ray studies of HPA lectin from the edible snail are reported. This protein is under development as a tool for early diagnosis of breast cancer.
Crystallization of a two-domain retroviral integrase is reported for the first time. The purification protocol describes a procedure that enables the isolation of this SIV integrase in large quantities.
Flavocetin-A, a high molecular mass C-type lectin-like protein from T. flavoviridis venom has been crystallized. The crystals belong to space group I4 with a = b = 121.0 and c = 63.2 Å and diffracted to at least 2.4 Å resolution.
Crystallization of human deoxyhaemoglobin in the complete absence of anions is reported. Crystals diffract to 1.87 Å and belong to space group P21212.
The hexameric glutamate dehydrogenase from T. profundus has been crystallized. The crystal contains the hexamer in its asymmetric unit and diffracts X-rays to 2.25 Å.
Crystallization, X-ray characterization and molecular-replacement solution of a Bowman–Birk type inhibitor from V. unguiculata are presented.
Crystallization and initial characterization of the ligand-binding domain of TyrR are described.
The pathogenesis-related protein of PR10 class from yellow lupin has been overexpressed in E. coli and crystallized. Diffraction data were collected to 2.45 Å resolution and the initial structure was solved by molecular replacement.
Guanidinoacetate methyltransferase is the enzyme that catalyzes the last step of creatine biosynthesis. Recombinant rat liver guanidinoacetate methyltransferase has been crystallized with guanidinoacetate and S-adenosylhomocysteine, and the crystals belong to the monoclinic space group P21 with unit-cell dimensions a = 54.8, b = 162.5, c = 56.1 Å, β = 96.8° at 93 K, and typically diffract beyond 2.8 Å.
Two crystal forms of malic enzyme from pigeon liver have been grown using hanging-drop vapour diffusion and preliminary X-ray diffraction data have been obtained.
Crystals of two quinocytochrome alcohol dehydrogenases, one monomeric and soluble and the other heterotrimeric and membrane-associated, have been obtained. Image-plate data from both proteins have been recorded and the location of a homologue of the monomeric protein was obtained by molecular replacement.
Phosphoenolpyruvate carboxylase, which is a key enzyme in the fixation of atmospheric CO2 in the C4 and crassulacean acid pathways, has been crystallized and preliminary X-ray diffraction data have been obtained.
Human cystatin C, a protein with amyloidogenic properies and a potent inhibitor of mammalian cysteine proteases, has been crystallized in two polymorphic forms: I432 (native protein and Se-Met derivatives) and P41212 (P43212), and in space group P42 after removal of ten N-terminal residues. The crystals of all the forms diffract to about 3 Å resolution and are characterized by multiple copies of the molecule in the asymmetric unit.
Crystals of the HincII restriction enzyme bound to cognate DNA have been grown. These crystals belong to the space group I222 (or I212121) and diffract to beyond 2.5 Å resolution.
The crystallization and preliminary characterization of 2-keto-3-deoxy-6-phosphogluconate from E. coli is reported. The structure of this enzyme will improve its utility in organic synthesis.
The conserved GTPase domain of the signal recognition particle from archaebacterium A. ambivalens has been cloned as a native protein and mutant and these have been crystallized and preliminary X-ray data have been obtained.
The α2 form of the E. coli HU protein crystallizes in space group I222. The two monomers are related by a twofold crystallographic axis.
Crystals of E. coli galactoside acetyltransferase have been prepared from solutions of ammonium sulfate containing acetyl-CoA and preliminary X-ray data have been obtained.
The N-terminal fragment of human protein disulfide isomerase containing a and b domains has been crystallized in two forms. Preliminary crystallographic investigations have been carried out.
Methods for purifying and crystallizing the precursors and autoprocessed enzymes of glycosylasparaginase are reported. The mutant precursors are active in autoproteolysis but have much slower rates.
Recombinant obelin crystallizes in relatively large single light-yellow hexagonal rods which show promising diffraction properties. A 1.8 Å data set has been collected and analysis of these data is in progress.


 journal menu
journal menu