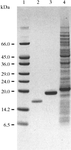
issue contents
January 2000 issue
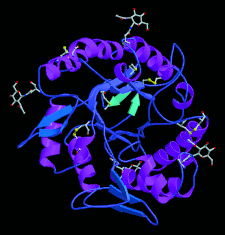
Cover illustration: Secondary structure of the T. reesei ![[beta]](/logos/entities/beta_rmgif.gif) -mannanase (p. 3).
-mannanase (p. 3).
editorial
Free 

international union of crystallography
Free 

research papers
The three-dimensional structure of the catalytic core domain of a T. reesei β-mannanase belonging to glycoside hydrolase family 5 has been solved and refined at 1.5 Å resolution. The starting phases were computed solely from the anomalous scattering of a single non-isomorphous platinum complex at 1.65 Å resolution.
The 2.0 Å structure of bovine interferon-γ; assessment of the structural differences between species
The structure of bovine interferon-γ, determined at 2.0 Å resolution by multiple isomorphous replacement, is compared with the other interferon-γ structures available.
PDB reference: bovine interferon-γ, 1d9c
The structure of a new neurotoxin BmK M2 from Chinese scorpion at 1.76 Å resolution has been studied with the sequence determined crystallographically. The structure further proved the existence of a non-proline cis peptide bond in the group III α-like scorpion toxins and showed the residues possibly involved in the high toxicity of BmK M2.
PDB reference: BmK M2, 1chz
The high-resolution refinement and analysis of the monomeric and dimeric forms of red abalone sperm lysin provides the basis for a model for the interaction of lysin with its egg receptor.
A program for inspection and interpretation of the Patterson function is described. Options are available for vector-based methods to locate heavy-atom sites, for finding sets from a list of possible heavy-atom positions and for checking of potential solutions.
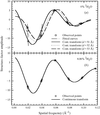
Neutron-diffraction data have been collected form 1,2-dioleoyl-sn-glycero-phosphocholine under conditions of increasing relative humidity at 0 and 8.06% 2H2O. These data are used to demonstrate the increased accuracy inherent in a real-time swelling-series method.
Good quality crystals of (Pro-Pro-Gly)10 were successfully grown in microgravity during the STS-95 mission.
crystallization papers
Human epidermal growth factor has been crystallized in trigonal and tetragonal space groups, and 3.0 Å resolution data have been collected for its trigonal crystals.
Crystallization and preliminary X-ray diffraction studies of N-utilizing substance-B (NusB) from M. tuberculosis are reported to 1.6 Å resolution.
Catabolite protein A from B. megaterium has been crystallized. The hexagonal crystals diffract to 2.55 Å resolution.
Crystals of a truncated form of the zymogen of the germination protease from spores of B. megaterium have been obtained by the vapor-diffusion method. The results of preliminary X-ray diffraction analysis of these crystals are discussed.
Full-length human recombinant m-calpain was crystallized in two crystal forms. A 2.3 Å data set was collected from a gold derivative by MAD techniques suitable for phase determination.
Recombinant adenosine kinase of T. gondii expressed in E. coli was purified, characterized and crystallized using ammonium sulfate or sodium sulfate at pH 8.5. These crystals belong to space group P21, with one molecule of protein in the asymmetric unit.
Two different crystal forms of cytochrome c6 from the red alga P. yezoensis have been obtained. Crystals belonging to space group P41212 or P43212 diffract to 1.8 Å resolution and crystals belonging to space group P212121 diffract to 1.6 Å resolution.
Lactate dehydrogenase from hyperthermophilic archaeon M. jannaschii has been crystallized. Its native data have been collected to 1.9 Å.
Ribosome recycling factor from E. coli has been crystallized and native data have been collected at 3.0 Å resolution using Cu Kα X-rays.
X-ray diffraction data have been collected from crystals of the fatty acid biosynthetic enzyme β-keto reductase from B. napus to a resolution of 2.8 Å and the crystal space group and cell parameters determined (P6422; a = b = 129.9, c = 93.1 Å, α = β = 90, γ = 120°). An initial molecular replacement solution has been determined using the structure of the enzyme hydroxysteroid dehydrogenase from Streptomyces hydrogenans as a search model.
Recombinant non-phosphorylating NAD+-dependent glyceraldehyde-3-phosphate dehydrogenase (GAPN) of the hyperthermophilic crenarchaeote Thermoproteus tenax has been overexpressed, purified and crystallized using the hanging-drop vapour-diffusion technique.
Crystals of the proteolytically truncated AhpF component of the alkyl hydroperoxide reductase from E. coli have been grown under oxidizing conditions. X-ray diffraction data, collected to 1.9 Å resolution using synchrotron radiation, are reported.
Attempts have been made to crystallize the primase from bacteriophage T4. A C-terminal fragment including residues 196–340 gave four crystal forms, one of which diffracts to 3.5 Å resolution.
Single crystals of a plant defence protein, the polygalacturonase-inhibiting protein (PGIP-1) from P. vulgaris, have been obtained. They belong to space group P21 and diffract to at least 2.3 Å resolution.
The crystallization of a type I chloramphenicol acetyltransferase using the concept of ionic strength reducers is reported.
Single crystals of an oligonucleotide construct containing triple helical DNA have been grown. These crystals diffract to 1.8 Å at a synchrotron X-ray beamline and contain bromuracil substitutions for thymine to permit MAD analysis.
short communications
A reason why some proteins crystallize best at supersaturation ratios of 2.5–3, rather than close to the supersolubility curve of their phase diagram, is proposed.
addenda and errata
Free 

Free 

international union of crystallography
Free 



 journal menu
journal menu