issue contents
July 2000 issue
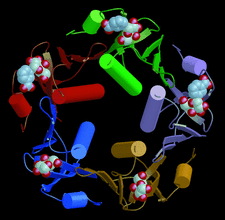
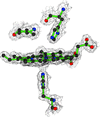
Cover illustration: Schematic diagram of the B pentamer of heat-labile enterotoxin, with each of the B subunits depicted in a different colour (p. 795).
scientific comment
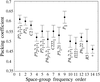
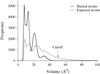
The packing coefficients (1 − solvent fraction) for 181 proteins have been calculated. The average packing coefficient is 57 ± 6% for all space groups. The most common space groups have higher packing coefficients compared with the less frequently occurring ones.
Protein densities calculated for buried atoms using the Voronoi algorithm show close agreement with those calculated for all atoms using the Connolly algorithm. These values are also in better agreement with experiment than those obtained recently by Andersson & Hovmöller.
research papers
Different binding modes were observed for m-carboxyphenyl-α-D-galactopyranoside in the five binding sites of the B pentamer of heat-labile enterotoxin.
PDB reference: PLT B5–MCPG, 1djr
Comparison of native haemoglobin and an artificial mutant shows that NCS breaks down around the site of the mutation.
The structural effect of genetically fusing the two α subunits in recombinant human hemoglobin is investigated. This genetic fusion between the C-terminus of one α subunit and the N-terminus of another, intramolecularly crosslinking the hemoglobin tetramer to prevent dissociation and thus rapid physiological clearance, is used in modifying recombinant hemoglobin to function as an oxygen-delivery therapeutic or `red blood cell substitute'.
The results of a statistical analysis of data in the BMCD suggest that different protein families show systematic biases in their patterns of successful crystallization. The software described here calculates Bayesian probabilities using data retrieved from an augmented version of the BMCD and uses these probabilities to bias the selection of data from an incomplete factorial design.
The design and implementation of the first World Wide Web-based system for macromolecular structural data deposition and validation is described. AutoDep's generic approach to data input may be easily adapted by other resources.
Analysis of refinement behavior through the eigenvalues and eigenvectors of the normal matrix.
For complexes of Ca, Mg, Mn, Fe, Cu and Zn in the Cambridge Structural Database, r.m.s. deviations of interbond angles from those of ideal geometry (octahedral, tetrahedral etc.) provide a measure of the distortion which can occur. Data is presented on interactions with fractional bond order which may have particular significance in carboxylate complexes of Zn.
Microgravity and ground-control chicken egg-white lysozyme crystals for IML-2 and LMS missions are compared, showing reduced mosaicity and larger uniform mosaic domains for the microgravity case but no differences in terms of strain. Enhanced-diffraction resolution is seen for microgravity over ground control for similar crystal volumes using synchrotron radiation.
High-resolution triple-axis X-ray diffraction has been applied to the characterization of the defect structure of hen egg-white lysozyme crystals.
Data were collected from the large unit cell crystals of the bacteriophage HK97 capsid using a third-generation synchrotron source. The resolution to which the data could be accurately measured increased from 5 to 3.5 Å.
crystallization papers
X-ray quality crystals have been obtained of recombinant human neutral endopeptidase (neprilysin) expressed in P. pastoris.
In order to investigate the biochemical antitumour activity of agglutinin, X-ray structure determination was exploited. Preliminary X-ray crystallographic results are presented.
Initial characterization of the substrate specificity along with the unit-cell parameters of enzyme crystals are reported.
The mitochondrial transcription factor sc-mtTFB from S. cerevisiae has been cloned, expressed, purified and crystallized.
Chondroitin sulfate ABC lyases are broad-specificity glycosaminoglycan-degrading enzymes. The crystallization of two isozymes of such a lyase produced by P. vulgaris is described.
Acutohaemolysin, a haemolytic toxin from A. acutus venom, has been characterized and crystallized. Preliminary X-ray diffraction analysis has been performed.
Crystallization of orotidine 5′-monophosphate decarboxylase from M. thermoautotrophicum with and without the inhibitor 6-azaUMP is reported.
Crystals of saccharopepsin from S. cerevisiae complexed with its natural inhibitor IA3 are reported. The crystals belonged to a hexagonal space group and diffracted to 5.0 Å
The thermostable sweet protein mabinlin II has been crystallized in two forms. One of them has been characterized as monoclinic and diffracts to 2.8 Å resolution.
CyaY, a 106-residue protein from E. coli, shows sequence similarity to human frataxin. Recombinant CyaY with a C-terminal His6 tag has been crystallized and diffraction data have been collected to 1.8 Å.
Crystals of the FERM domain of radixin, a protein crosslinking plasma membranes and actin filaments, were obtained. Crystals belong to space group P41212 or P43212 (a = b = 96.36, c = 133.16 Å) and diffract to a resolution of 3.0 Å.
Aminotripeptidase (peptidase T) from S. typhimurium has been crystallized. Crystal quality was improved by the use of a His-tag derivative purified in a single step. X-ray diffraction data to 2.6 Å have been collected on native and selenomethionine protein and the Se-atom positions have been determined.
Carbonic anhydrase from garden pea has been purified to homogeneity and crystallized. The orthorhombic crystals most probably contain a native octamer per asymmetric unit.
The EVH1-domain of Vesl-2b, a neuronal protein involved in synaptic plasticity and long-term potentiation, has been crystallized. Crystals diffract to 2.4 Å resolution.
short communications
A completely ordered detergent molecule is revealed in the 1.3 Å resolution structure of the retinoic acid receptor. A crystal contact generates a binding site of appropriate size for a dodecyl-α-D-maltoside molecule.
addenda and errata
Free 

An erratum to the paper by Guo et al. (2000) Acta Cryst. D56, 451–457.


 journal menu
journal menu