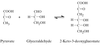issue contents
November 2000 issue
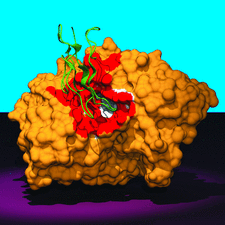
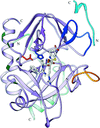
Cover illustration: Human acetylcholinesterase complexed with fasciculin-II. hAChE is shown as a solvent-accessible surface in gold and FAS-II is shown as a ribbon in green. The red area corresponds to points on the surface ![[less-than or equal to]](/logos/entities/le_rmgif.gif) 4 Å from FAS-II and the inner surface of the active-site gorge is coloured grey (p. 1385).
4 Å from FAS-II and the inner surface of the active-site gorge is coloured grey (p. 1385).
research papers
The gene encoding iron superoxide dismutase from P. ovalis has been cloned and sequenced; the recombinant enzyme has been crystallized and its structure determined.
PDB reference: P. ovalis iron superoxide dismutase, 1dt0
The structure of a highly thermostable family II xylanase has been determined at 1.8 Å resolution. Thermostability is attributed to increased polar surface and increased hydrogen bonding.
PDB reference: XynB, 1f5j
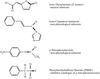
The structure of the DLH (C123S) with PMS bound indicates that the reason the enzyme is able to catalyse a wide range of dissimilar substrates is a consequence of its capacity to undergo a coordinated restructuring of the active site and associated regions of the molecule without compromising the stability of its tertiary fold.
PDB reference: DLH C123S mutant–PMSF complex, 1ggv
IUCr article reference: gr2044
The structures of recombinant wild-type human acetylcholinesterase and of its E202Q mutant, as complexes with fasciculin-II, a `three-finger' snake neurotoxin, have been determined to 2.8 and 2.7 Å resolution, respectively. The overall structures of these complexes are similar to those of the Torpedo and mouse enzymes with fasciculin-II. Comparison of the wild-type and the mutant structures shows that removal of the charged group from the protein core, and its substitution by a neutral isosteric moiety, does not disrupt the functional architecture of the active center.
The three-dimensional structure of the ternary complex consisting of human α-thrombin, hirugen and the active-site inhibitor RWJ-51438 has been determined at 1.7 Å resolution.
PDB reference: α-thrombin–RWJ-51438 complex, 1doj
The structure of bucandin, a novel three-finger toxin, has been determined by direct methods. The full-matrix least-squares refinement to atomic resolution, with anisotropic temperature factors, converged to an R factor of 12.4%.
PDB reference: bucandin, 1f94
The first structure of a complex between ferredoxin-NADP+ reductase and ferredoxin has been solved at 2.4 Å resolution by molecular replacement, anomalous dispersion and Rmin search methods. The crystal was treated as a merohedral twin.
Analyzing the anomalous diffraction from seven different selenomethionine-labelled crystals, we show that the combination of single-wavelength anomalous-diffraction phasing and solvent flattening is sufficient to unambiguously determine most structures.
A systematic analysis of local and global protein electron-density map properties is used to derive an empirical function to measure the average error in diffraction phase sets.
crystallization papers
Slow evaporation of the reservoir in microdialysis can lead to a significant increase in the crystal size of proteins for which the crystallization region of supersaturation is particularly limited.
The acidic cytochrome c4 from T. ferrooxidans has been cloned, expressed, purified and crystallized. The MAD method using the four Fe per asymmetric unit is used to solve the protein structure.
The crystallization and preliminary X-ray analysis of a mastoparan peptide isolated from the venom of the solitary wasp A. flavomarginatum micado is reported.
The growth of three crystal forms of 2-keto-3-deoxygluconate aldolase from the hyperthermophilic archaeon S. solfataricus is reported.
The first crystallization of a serpin that is either viral or a cysteine proteinase inhibitor is described.
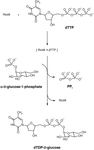
Glucose dehydrogenase from B. megaterium IWG3 has been crystallized and 1.7 Å resolution data have been collected using a synchrotron-radiation source.
The key response regulator of light regulation, Rcp1, from Synechocystis sp. has been crystallized in a form suitable for X-ray diffraction studies. The crystals belong to space group P63 and diffracted to 2.5 Å.
The lethal factor (LF) produced by toxigenic strains of B. anthracis is a Zn-endopeptidase responsible of the lethal effects of anthrax. LF has been expressed in E. coli as recombinant GST fusion, purified and crystallized; preliminary crystallographic studies are described.
Crystals of a functional form of pneumolysin, a virulence factor of S. pneumoniae, have been obtained in complex with its activator, a cholesterol molecule, by the vapor-diffusion method. The crystallization process as well as the preliminary X-ray analysis are discussed.
The DNA-binding domain of the MerR family member MtaN has been crystallized in space group I212121. Intensity data have been collected from these crystals at 100 K at a synchrotron-radiation source to 2.75 Å resolution.
Annexin I, a member of the annexin family of Ca2+- and phospholipid-binding proteins, has been crystallized with the complete N-terminus.
A bacterial lysozyme produced by S. globisporus was crystallized in space group P41212, with unit-cell parameters a = 63.11, c = 121.1 Å. A clear molecular-replacement solution was obtained and the structure refinement is in progress.
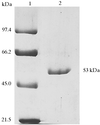
Full-length human tau-protein kinase I (TPK-I; also known as glycogen synthase kinase-3β, GSK-3β) was produced in E. coli and crystallized. The crystals diffract up to 2.3 Å resolution and belong to the orthorhombic space group 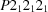 , with unit-cell parameters a = 82.9, b = 86.1, c = 178.1 Å measured at 100 K.
, with unit-cell parameters a = 82.9, b = 86.1, c = 178.1 Å measured at 100 K.
Trichomaglin has been crystallized in two crystal forms.
The membrane-external domain of the immune modulator CTLA-4 (CD152) has been crystallized; the crystals diffract to 2.7 Å resolution at synchrotron sources.
F. pennivorans Ven5 pullulanase type I was crystallized in the presence of the inhibitor β-cyclodextrin. The crystals belong to space group P212121, with unit-cell parameters a = 76.8, b = 96.2, c = 98.5 Å, diffract to 2.2 Å using synchrotron radiation and contain one monomer in the asymmetric unit.
Native and selenomethionyl-labelled RNA-dependent RNA polymerase from bacteriophage Φ6 crystallizes in space groups P21 and P31/P32. Data have been collected for both space groups to better than 3 Å resolution.
Monoclinic crystals of the maize auxin receptor ABP1 diffract to 1.9 Å resolution.
Preliminary crystal data have been obtained for the first time for human CCG1-interacting factor B. The results of preliminary X-ray diffraction are discussed.
Preliminary crystal data have been obtained for a new crystal form of human secretory phospholipase A2 IIA crystallized with pentapeptide inhibitors. The enzyme crystallizes in space group C2, with unit-cell parameters a = 140.8, b = 38.9, c = 109.1 Å, β = 125.1°.
Nucleoside diphosphate kinase from M. jannaschii has been crystallized using polyethylene glycol 4000 as precipitant. Native data have been collected to 2.30 Å resolution using synchrotron X-rays.
Crystals of ATP-phosphoribosyltransferase from E. coli were obtained by the vapour-diffusion method and diffract to 2.7 Å. The quaternary structure is determined based on the preliminary crystallographic studies.
Here the crystallization, data collection and molecular-replacement solutions obtained for two crystalline forms of triose phosphate isomerase, one determined at 85 K and the other at 298 K are reported.
Dihydroxybutanone phosphate synthase from M. grisea was cloned, expressed and crystallized. Crystals diffract to 1.0 Å resolution.
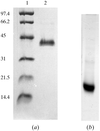
Oligandrin, a small 10 kDa phytotoxin with sterol-carrier properties, has been crystallized from PEG 4000 at pH 4.5.
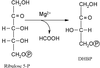
The anabolism of L-rhamnose in many pathogenic bacteria presents itself as a possible novel therapeutic target. RmlA is the first of the four enzymes involved in this pathway. Crystallization and preliminary structural characterization are reported.
A new E. coli L-asparaginase belonging to the class of Ntn hydrolases has been crystallized in the orthorhombic space group P212121. The crystals diffract to 1.65 Å resolution and contain an (αβ)2 heterotetramer in the asymmetric unit, where the subunits α and β are the product of autocatalytic cleavage of the immature protein.
β-galactosidase from Penicillium sp. was crystallized and diffracted to 1.85 Å resolution. It belonged to the tetragonal P43(P41) space group, with unit-cell parameters a = b = 110.82, c = 161.28 Å.
meeting reports
Free 

Report of the workshop Toward Automation of Structure Determination in Macromolecular Crystallography held on 20–21 June 1999.
addenda and errata
Free 

Free 



 journal menu
journal menu