issue contents
January 2001 issue
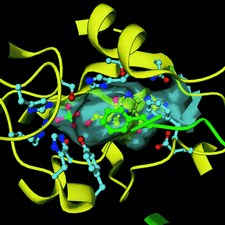
Cover illustration: Binding pocket of calf spleen PNP described through the solvent-accessible surface calculated without phosphate and inhibitor (p. 30).
research papers
In the crystal structure of human erythrocyte catalase, the N- and C-terminal segments corresponding to the first and last exons of the gene were not located in the tetragonal I41 form. Two similar types of lattice contacts between the HEC molecules were related by a pseudo-4122 symmetry.
PDB reference: human erythrocyte catalase, 1f4j
The paper presents results of atomic resolution X-ray studies of the F. oxysporum trypsin with its own autolysis products and in the presence of the added amino acids arginine, lysine and glutamate to investigate their binding in the S1 specificity pocket.
PDB references: TRY-N, 1gdu; TRY-F, 1gdq; TRY-ARG, 1fn8; TRY-LYS, 1gdn; TRY-LYS2, 1fy5; TRY-GLN, 1fy4
The structure of human EF-hand calcium-binding protein S100A12 in its calcium-bound form has been determined to 1.95 Å resolution. Comparison between members of the S100 family suggests the location of the target-binding region of S100A12 and provides a better understanding of the role of the residues important for intra- and inter-subunit hydrophobic interactions.
PDB reference: S100A12, 1e8a
The crystal structure of the ternary complex of calf spleen purine nucleoside phosphonylase (PNP) with an N(7)-acycloguanosine inhibitor and phosphate shows the guanine base in an inverted orientation and the acyclonucleoside chain in a folded conformation. This new binding mode accounts for the observed substrate properties of 7-β-D-ribonuclosides, and also provides a new lead for the design of PNP inhibitor.
PDB reference: PNP ternary complex, 1fxu
The crystal structure of T. fusca β-mannanase has been solved despite the absence of the major part of the amino-acid sequence. A high-quality electron-density map provided the necessary information to obtain the corresponding DNA sequence.
PDB reference: β-mannanase, 1bqc
The structure of the CheY–CheA124–257 complex has been determined in three conditions: the native form, after soaking with a phosphorylating agent and after soaking with an inhibitor of the phosphorylation reaction. According to the geometry of the CheY active site, these structures illustrate different functional states of the response regulator.
The crystal structure of R-phycoeythrin has been determined in the R3 space group from a twinned crystal using SHELX97. The 2.2 Å resolution refined model shows good stereochemistry; the data also allowed the proposal of the presence of a helical segment of linker protein in the central channel of R-PE.
PDB reference: R-phycoerythrin, 1eyx
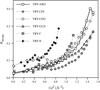
The dynamic response of tetragonal lysozyme crystals to dehydration shows the abruptness, metastability and hysteresis characteristic of a structural transition. The success of more general post-growth crystal treatments may rest on either avoiding lattice transitions, minimizing disorder created during such transitions or maintaining the lattice in an ordered metastable state.
Improved thermal models that include convection are developed which replace Helliwell's adiabatic approximation. Temperature rises of 6 K are calculated for a cryocooled 100 µm thick crystal and of 18 K for a room-temperature air-cooled 1 mm thick crystal for an 8 keV 1013 photons s−1 mm−2 beam. The importance of internal heat conduction within the crystal is also carefully examined.
A number of real-space torsion-angle refinement techniques have been implemented in a model-building program providing near-automation for macromolecular structure tracing and model building.
The low-density elimination method has been improved and tested for ab initio solutions of several structures including proteins.
The problem of calculating the structure-factor amplitudes of the anomalously scattering atoms within a protein crystal is considered.
A preliminary crystallographic model for the low-density lipoprotein (LDL) particle was obtained at a resolution of 27 Å using several ab initio phasing methods.
The implementation in the program REFMAC of anisotropic refinement with a small number of parameters via the TLS parameterization is described. Sample results are given for two examples, which show improvements in the free R factor of several percent and an improved description of NCS-related molecules.
crystallization papers
Crystals of the recombinant form of a glutathione S-transferase from A. gambiae, agGST1-6, have been grown in at least five different crystal forms with a broad range of effective resolution.
The first crystals from a chemosensory protein, CSP2, have been obtained by the hanging-drop vapour-diffusion method.
A growth-promoting β-glucosidase in plants, Zm-p60.1 from maize, has been overexpressed, purified and crystallized. Several crystal forms have been observed, one of which diffracts to better than 2.0 Å.
Monoclinic crystals of N. alata SF11-RNase have been obtained. Native data have been collected to 1.55 Å resolution using the SPring-8 synchrotron-radiation source.
(R)-Hydratase from A. caviae involved in PHA biosynthesis has been crystallized in space group C2, with unit-cell parameters a = 111.54, b = 59.29, c = 47.27 Å, β = 113.04°.
The `squid-type' DFPase from L. vulgaris has been recombinantly expressed and crystallized. The orthorhombic crystals diffracted beyond 2.0 Å resolution using a Cu Kα radiation source.
A histidine-tagged form of 5′-methylthioadenosine/S-adenosylhomocysteine nucleosidase from E. coli has been overexpressed and crystallized. A native data set, complete to 2.3 Å has been collected.
Dihydropyrimidine dehydrogenase catalyzes reduction of pyrimidine bases to the corresponding 5,6-dihydro derivatives, the first step in the pyrimidine catabolic pathway. Crystals of this enzyme diffract to at least 2.5 Å and belong to space group P21 (unit-cell parameters a = 82.0, b = 159.3, c = 163.6 Å, β = 96.1°).
The large extracellular domain of CD81, a member of the tetraspanin family and a receptor protein for hepatitis C virus envelope E2 glycoprotein, has been expressed, purified and subsequently crystallized using the sitting-drop vapour-diffusion technique.
The yeast translation elongation factor complex eEF1A–eEF1Bα has been crystallized and data collected to 1.67 Å. The phases were determined from a three-wavelength MAD experiment with three selenomethionines in the 61 kDa asymmetric unit.
Expression, purification and crystallization of class-C β-lactamase from E. cloacae 908R and two point mutants (S64C and S84C) are reported together with the preliminary X-ray analysis.
Crystals of a S305C GlyDH mutant were obtained by the hanging-drop vapour-diffusion method, using ammonium sulfate and PEG 400 as precipitating agents, in the presence and absence of NAD+. The crystals belong to space group I422, with approximate unit-cell parameters a = b = 105, c = 149 Å and one subunit in the asymmetric unit, corresponding to a packing density of 2.6 Å3 Da−1.
An intact human immunoglobulin with a full-length hinge has been crystallized for the first time in a form in which all of the Ig domains are ordered.
Monoclinic crystals of Japanese pear S3-RNase have been obtained. Native data have been collected to 1.5 Å resolution using the SPring-8 synchrotron-radiation source.
international union of crystallography
Free 



 journal menu
journal menu