issue contents
June 2001 issue

Cover illustration: An atomic force micrograph of an array of screw dislocations and stacking faults on the developing surface of a twinned rhombohedral crystal of the plant seed protein canavalin (p. 829).
research papers
Derivatization with a mercury compound increases the diffraction limit of E. coli dUTPase crystals to atomic resolution. Data collection to 1.05 Å allows the structure to be solved from the position of the single Hg atom in the asymmetric unit. The final refined dUTPase derivative structure is described and compared with the native structure.
The structure of bar-headed goose aquomet haemoglobin showed quaternary structural differences from lowland animal haemoglobins; critical residue differences are located in the α1β1 interface.
PDB reference: bar-headed goose aquomet Hb, 1c40
The monoclinic crystal structure of C-phycocyanin from S. platensis has been determined at 2.2 Å resolution. The unique side-by-side packing of two hexamers in the asymmetric unit makes it possible that lateral energy transfer takes place by the β155–β155 route.
PDB reference: C-phycocyanin, 1gh0
The crystal structure of the N-terminal regulatory domain of the Y. pestis effector protein YopH has been determined at a resolution of 2.0 Å.
PDB reference: YopH, 1huf
The structure of a homohexameric protein particle was solved by employing a tantalum bromide derivative of the crystals obtained. A low-resolution MAD experiment (three wavelengths) rendered phases that could be improved by averaging and extended by applying density modifications.
The order/disorder changes of active-site loops upon substrate binding are described and implications for substrate recognition and stabilization are shown, along with the effect of a higher crystallization pH.
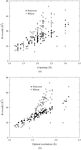
The program SFCHECK, which analyses the quality of the structure-factor data, and the agreement of those data with the atomic coordinates, is used to survey 105 nucleic acid crystal structures from the NDB.
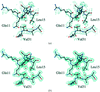
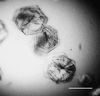
Hemihedrally twinned rhombohedral crystals of the plant seed protein canavalin were successfully refined. Their investigation by atomic force microscopy revealed an extensive defect structure that is likely to be a consequence of the twinning.
Laboratory double-axis multiple-crystal X-ray topography and triple-axis high angular resolution reciprocal-space mapping have been combined to investigate the influence of acetylated lysozyme upon lysozyme crystal perfection.
crystallization papers
Crystallization of human muscle fructose-1,6-bisphosphatase has yielded complete data to 2.04 Å.
AML1/Runx-1/CBFα(Runt)–CBFβ–C/EBPβ(bZip)–DNA, AML1/Runx-1/CBFα(Runt)–C/EBPβ(bZip)–DNA and AML1/Runx-1/CBFα(Runt)–DNA, three types of protein–DNA complexes, have been crystallized. The former two crystals diffracted to 3 Å resolution and the latter crystal diffracted to 2.65 Å resolution.
Two kinds of C-terminal fragments of human C/EBPβ (residues 259–345 and 259–336) were crystallized in complex with a 16 bp DNA fragment from the tom-1 promoter. These two types of crystals diffracted to 2.1 and 1.8 Å resolution, respectively.
Two different crystal forms of meso-2,3-butanediol dehydrogenase from K. pneumoniae IAM1063 have been obtained. Crystal forms I and II belong to space groups C2 and P21 and diffract to 2.0 and 1.7 Å resolution, respectively.
Rhodnius haem-binding protein (RHBP) from the bloodsucking insect R. prolixus, a 15 kDa protein, has been crystallized using polyethylene glycol as a precipitant. The crystals belong to the space group P41(3)212 and diffract beyond 2.6 Å resolution.
Sorcin, a penta EF-hand Ca2+-binding protein, and its Ca2+-binding domain have been crystallized. The crystals of the entire molecule diffract poorly, but two crystal forms grown from the domain that lacks the first 32 amino acids diffract to 2.1 and 2.7 Å resolution.
Crystals of ribosomal protein L30e from the hyperthermophilic archaeon T. celer were obtained and data diffracting to 1.96 Å were collected using a Cu Kα X-ray source. The crystals belong to space group P61/P65, with unit-cell parameters a = b = 48.32, c = 86.42 Å.
Met8p was successfully crystallized by combining crystallization screening results with incremental adjustments to the protein storage buffer.
Two glutathione S-transferase isozymes from the mosquito Anopheles dirus (AdGST1-3 and AdGST1-4) from an alternately spliced gene family have been expressed, purified and crystallized. Determination of these structures will advance our understanding of how these enzymes inactivate pesticides and the structural consequences of alternate splicing.
The space group and the unit-cell parameters of crystals of C2A2C4A2 and Patterson and molecular-replacement studies on the crystals suggest that the molecules assume a tetraplex structure.
Four different crystal forms of B. subtilis dUTPase in unliganded and potentially liganded (with a substrate analogue) forms have been analysed using synchrotron radiation.
NADP-dependent glyceraldehyde-3-phosphate dehydrogenase of Synechococcus PCC 7942 was crystallized in two different forms using ammonium sulfate as a precipitant. Form I crystals belong to the hexagonal space group P65 and form II crystals belong to the monoclinic space group C2.
S100A1, a Ca2+-binding protein from the S100 protein family, has been crystallized. The crystals were stable during exposure to X-rays and diffracted to 2.6 Å resolution in-house.
Hexahistidine-tagged AlgS of Sphingomonas sp. A1 was overexpressed in E. coli and crystallized. X-ray diffraction data to 3.2 Å have been collected from the native crystal.
The Lrp-like regulatory protein, LrpA, from P. furiosus has been crystallized in a form suitable for X-ray diffraction studies. An octameric assembly for the protein is suggested by its behaviour on gel-filtration columns and analysis of the self-rotation function.
Crystals of RhoGDI complexed with the FERM domain of radixin were obtained and belong to the space group P21212, with unit-cell parameters a = 130.9, b = 151.2, c = 71.2 Å. Diffraction data to a resolution of 2.9 Å have been obtained using synchrotron radiation.
Crystals of the complex between the FERM domain of radixin and the cytoplasmic tail of ICAM-2 were obtained. The binary complex crystals were found to belong to the space group P3121 or P3221, with unit-cell parameters a = b = 100.44, c = 99.49 Å, and diffract to 2.6 Å resolution.
A 38.5 kDa cytoplasmic domain of Sec12p from S. cerevisiae, a guanine nucleotide-exchange factor of a GTP-binding protein Sar1p, a key factor in vesicle transport from the endoplasmic reticulum to the Golgi apparatus, has been expressed in S. cerevisiae and in E. coli. The proteins have been purified to homogeneity and X-ray diffraction data sets have been collected from two crystal forms to 2.6 and 2.0 Å resolution.
The cell-division inhibitor MinD from P. horikoshii was crystallized and preliminary X-ray diffraction studies were performed.
The receptor-binding domain of human B7-2 and the disulfide-linked homodimer of human CTLA-4 have been produced in E. coli, purified and cocrystallized in a monoclinic form (space group P21). A single crystal was obtained that diffracted to 3.2 Å resolution.
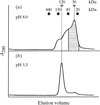
The crystallization conditions of a recombinant form of the complete sequence of human γ-interferon have been determined after studying the behaviour of this protein in solution by small-angle X-ray scattering (SAXS) as a function of pH and salt type.
Recombinant catechol-O-methyltransferase has been crystallized with a new tight-binding inhibitor. This enzyme is an important target in the treatment of Parkinson's disease.
The E. coli Hsp100 ClpB nucleotide-binding domain 1 (NBD1) has been cloned, purified and crystallized. The crystals diffracted to 1.8 Å.
Snake gourd seed lectin has been crystallized. Preliminary X-ray studies indicate the lectin to be homologous to type II ribosome-inactivating proteins.
Crystals of T. cruzi dUTPase in two crystals forms, both belonging to the hexagonal space group P6322, have been obtained. Form I is similar to crystals reported previously, but the crystals are of better quality and produced under different conditions, while form II has not been reported before.
Glycerol 3-phosphate cytidylyltransferase from S. aureus has been cloned, expressed, purified and subsequently crystallized. Determination of crystallization conditions required a combination of three optimization strategies.
short communications
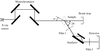
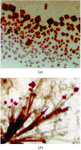
Low-pH incubation of human embryonic haemoglobin Portland (ζ2γ2) has resulted in the preferential crystallization of haemoglobin Bart's (γ4), as the result of dissociation of the embryonic heterotetramer and preferential formation of the homotetramer.
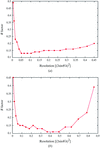
The effect of the amphiphile heptanetriol on the critical micelle concentration and the cloud-point temperature was measured for several detergents commonly employed in the crystallization of membrane proteins.


 journal menu
journal menu