issue contents
September 2002 issue
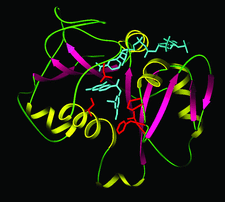
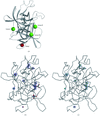
Cover illustration: Model of the tertiary structure of the Pneumocystis carinii DHFR-NADP+-3',5'-dimethoxypyrido[2,3-d]pyrimidine ternary complex. Helices are in yellow, sheets are in red and loops are in green, p. 1393.
research papers
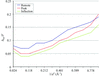
The parameters of the flat bulk-solvent model have been studied. A procedure for their search is suggested which allows improvement of the correspondence between the calculated and experimental data.
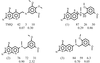
The crystal structure studies of two ternary complexes of P. carinii dihydrofolate reductase with the cofactor NADP+ and potent antifolates, the N9—C10 reversed-bridge inhibitor 2,4-diamino-6-[N-(2′,5′-dimethoxybenzyl)-N-methylamino]quinazoline and its 3′,5′-dimethoxypyrido[2,3-d]pyrimidine analog, have been carried out.
The crystal structure of cyclized green fluorescent protein (cGFP), which was engineered using split-intein technology, has been refined and shows that the linker does not impose any restraints on the fold. A new mode of dimerization of GFP was observed.
PDB reference: cyclized green fluorescent protein, 1kp5, r1kp5sf
Orthorhombic crystals of porcine pancreatic elastase have been grown in the presence of sodium sulfate and in the presence of sodium citrate plus calcium chloride. The observed differences in the diffraction patterns lead to the conclusion that elastase in its canonical Na2SO4 crystal form does not contain calcium in its metal-binding site but sodium instead.
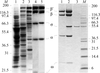
Crystals derived by soaking in bromide solutions can subsequently be derived under a xenon atmosphere. Intensity data collected to lower energy than the bromine absorption edge can be used to determine the xenon position and the resultant phase information used to determine the bromine substructure from data collected to higher energy than the bromine absorption edge.
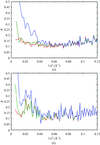
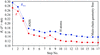
Crystallographic normal-mode refinement was carried out for the diffraction data of human lysozyme obtained at temperatures ranging from 113 to 178 K. The effects of a dynamic transition that occurred at about 150 K on the dynamic structures as well as the static structures of a protein molecule in a crystal are discussed.
The structure of the endoglucanase A from Clostridium thermocellum CelA has been re-solved by three-wavelength MAD.
PDB reference: endoglucanase A, 1is9, 1is9sf
The phytotoxic cryptogein, a 10 kDa protein from an oomycete fungus, gives a cholesterol complex at high resolution.
PDB reference: cryptogein–cholesterol complex, 1lri, r1lrisf
The 1 Å resolution structure of a mutant of trypsin inhibitor from squash reveals a 4:1 protein–zinc complex with unusual coordination.
PDB reference: squash trypsin inhibitor, 1lu0, r1lu0sf
crystallization papers
Comparison of the structure of the adenovirus proteinase, whose crystallization and X-ray diffraction are reported here, with that of the enzyme activated by the 11-amino acid peptide pVIc will reveal why the enzyme is inactive and how the enzyme is activated by pVIc. The low solvent content, 29.2%, may be the reason why this crystal diffracted to such high resolution, 0.98 Å.
Cold-active protein-tyrosine phosphatase of a psychrophile, Shewanella sp., has beencrystallized and the diffraction data were collected using Cu Kα X-ray radiation.
Peptide methionine sulfoxide reductase B from N. meningitidis and its selenomethionine-substituted form were crystallized. A MAD experiment was performed at 1.8 Å resolution and an electron-density map of excellent quality was obtained from the resulting phases.
The R-2-hydroxypropyl-coenzyme M dehydrogenase has been crystallized in the presence and absence of its reaction substrates and 1.90 Å resolution data have been collected.
The 65 kDa heat-shock protein (Cpn60-2) from M. tuberculosis has been crystallized and its preliminary crystallographic characterization is reported.
Crystallization and preliminary crystallographic study of the MntC manganese-binding protein, the solute-binding component of an ABC transport system from Synechocytis sp. PCC 6803, is reported.
Pyridoxal kinase from sheep brain has been crystallized in space group P212121; the crystals diffract to 2.1 Å. Two heavy-atom derivatives have been prepared.
The first D-aminoacylase crystal from A. faecalis has been obtained in hanging drops at pH 5.6 by the vapour-diffusion method using 30% polyethylene glycol 4000 as precipitant.
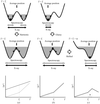
T4 phage β-glucosyltransferase has been crystallized with an abasic site-containing DNA and uridine diphosphoglucose to investigate a base-flipping phenomenon.
The endolysin coded by the phage Cp-1 has been crystallized by the hanging-drop vapour-diffusion method and by addition of a specific detergent. Native data have been collected to 2.1 Å resolution.
The crystallization of a recombinant CotA protein produced in E. coli and the preliminary characterization of the crystals is reported
Crystals were obtained of a cold-adapted xylanase and both native data and SAD data from a SeMet-substituted enzyme were collected.
Bacterial RNA polymerase holoenzyme, an assembly of six protein subunits with a total molecular mass of ∼450 kDa, was crystallized under near-physiological conditions. A complete diffraction data set was collected at 3 Å resolution. The structure determination and refinement is in progress.
The wild-type peroxiredoxin from poplar and the V152C mutated protein have been crystallized. Native data sets, X-ray analyses and a molecular-replacement solution are presented.
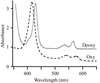
The direct oxygen sensor (DOSH) protein from E. coli is uniquely six-coordinate in both ferric and ferrous heme states and the oxidized form of this protein has been crystallized.
Betaine-homocysteine S-methyltransferase from rat liver has been purified and crystallized. Diffraction data from native, selenomethionine-labelled and two heavy-atom derivatives have been collected using synchrotron sources.


 journal menu
journal menu