issue contents
May 2003 issue
Cover illustration: Atomic displacement parameters (50% ellipsoids) for a streptavidin subunit containing an Arg-Gly-Asp (RGD) sequence from fibronectin (p. 828).
research papers
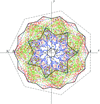
Splitting the heptameric chains in the GroEL–GroES chaperonin complex into segments gives rise to molecular forms which obey a law of rational indices and which are invariant with respect to scale-rotations with integral entries.
The molecular forms of heptameric chain segments of the GroEL–GroES chaperonin complex lead to folding points at form boundaries. Examples are given of such Cα positions related by integral scale-rotations.
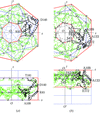
A new recursive phasing method has been developed and used to produce an electron-density map of lysozyme, based on a unique phase-occurrence pattern in a triplet-phase data set measured by reference-beam diffraction.
A new crystal form of the hexameric DNA helicase RepA encoded by plasmid RSF1010 that has sulfate anions bound to the active sites has been determined to 1.95 Å resolution. These structural data together with electron-microscopy studies implicate conformational changes that lead towards an `open' stucture of RepA upon binding of nucleoside 5′-triphosphates.
PDB reference: RepA–sulfate complex, 1nlf, r1nlfsf
A database of 41 experimentally phased electron-density maps between 1.3 and 2.9 Å resolution was used to evaluate the performance of the automated protein model-building procedures within the ARP/wARP, MAID and RESOLVE programs.
Cell-adhesion properties can be engineered into streptavidin by the insertion of selected fibronectin or osteopontin sequences into an external loop. High-resolution crystal structures of two such streptavidins show that the loops further sample the conformations seen in other RGD motifs.
The structure of a glyceraldehye 3-phosphate dehydrogenase from a denitrifying bacterium has been determined at 1.7 Å resolution. It confirms the essential role of Cys154 in the enzyme's activity.
PDB reference: glyceraldehyde 3-phosphate dehydrogenase, 1obf, r1obfsf
The bound water structure in medium–high versus high-resolution protein (Fo − Fc) difference electron-density maps yields more detail than (2Fo − Fc) maps. Tests on truncated resolution cases and `real' data-set cases are examined. Re-refinements at 3.2 Å show distances that can agree with known values but B values that do not agree with known values.
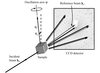
Low-resolution ab initio images obtained from crystals of a RNA–protein complex allow the protein and RNA molecules to be seen alone or in complex depending on the diffraction density of the bulk solvent.
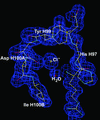
The structure of S100A12, a member of the S100 family of proteins, has been determined in complex with copper at 2.19 Å resolution. Copper binds to the same site that zinc binds to in the structure of S100A7. The data suggest that copper binding may be essential for the function of S100A12 and probably other calgranulins S100A8 and S100A9 in the early immune response.
PDB reference: S100A12–copper complex, 1odb, r1odbsf
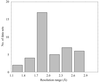
Four individual data sets collected at various wavelengths were used for SAD phasing of two crystal forms of glucose isomerase, including one previously undescribed, using the anomalous signal of a single Mn2+ ion present in the 44 kDa molecule, which ranged from 1.2 to 0.6%.
PDB reference: P21212 glucose isomerase, 1oad, r1oadsf
The structure of the GMPPNP-bound constitutively active Q63L mutant of the cytosolic RhoA GTPase was solved at 1.55 Å resolution and refined using anisotropic displacement parameters. The structure is similar to the Val14 mutant and cannot explain the functional differences that are observed in vivo.
PDB reference: Q63L RhoA mutant, 1kmq, r1kmqsf
RNA polymerase-associated stringent starvation proteins A from E. coli, Y. pestis, V. cholerae and P. aeruginosa were characterized and X-ray diffraction-quality crystals were obtained for the Y. pestis protein only.
The phasing and ab initio structure-determination potential of organic arsenic derivatives have been studied using lysozyme crystals.
PDB reference: hen egg-white lysozyme, arsenic derivative, 1n4f, r1n4fsf
The structure of aminopeptidase P from E. coli in the orthorhombic space group C2221 has six subunits in the asymmetric unit. The molecular structure is compared with those previously reported in hexagonal and tetragonal space groups.
PDB reference: orthorhombic AMPP, 1m35, r1m35sf
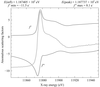
A computational method to partially correct radiation damage to protein crystals is described and applied to a test data set with a weak anomalous signal. It is shown that the correction results in an enhancement of SAD phases and electron-density maps.
crystallization papers
R88A, a mutant of restriction endonuclease EcoRII, has been crystallized in a different crystal form to that of the wild type. The mutant crystals diffract to 2.1 Å resolution.
This study describes the first crystallization and preliminary X-ray analysis of the α-L-arabinofuranosidase from G. stearothermophilus T-6, a family 51 glycoside hydrolase. Purified native and selenomethionine-containing enzymes were crystallized in two different space groups, P21 and R3. The R3 crystal form diffracted X-rays to a resolution of 1.8 Å.
An antennal protein from the cockroach L. maderae, expressed specifically in the female adult antennae, shares all the hallmarks of the PBP family. It has been expressed as recombinant protein in E. coli periplasm and crystallized by the sitting-drop vapor-diffusion method using a nanodrop dispensing robot.
The crystallization of a PBP from the honeybee antennae expressed as recombinant protein in the yeast P. pastoris is reported. Unique crystals of this PBP have been obtained with the sitting-drop vapor-diffusion method using a nanodrop dispensing robot with a novel optimization procedure.
Two forms of two-dimensional crystals composed of monomeric CRP have been obtained on negatively charged lipid monolayers: a previously reported form, MI, and a new form, MII.
YPD1, a histidine-containing phosphotransfer (HPt) protein, has been co-crystallized with its upstream phosphodonor, the SLN1-R1 response-regulator domain.
Ribosomal protein L11 methyltransferase from T. thermophilus HB8 has been crystallized. X-ray diffraction data were collected to 1.9 Å using synchrotron radiation.
Human flap endonuclease-1 complexed with proliferating-cell nuclear antigen has been crystallized. A diffraction data set was collected to a resolution of 3.0 Å.
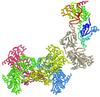
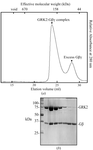
A complex between G protein-coupled receptor kinase 2 (GRK2) and Gβ1γ2 has been crystallized in space group C2, with unit-cell parameters a = 187.0, b = 72.1, c = 122.0 Å, β = 115.2°. Crystals diffract to beyond 3.2 Å spacing with Cu Kα radiation.
Crystallization studies on the domain of the α(I) subunit of human collagen prolyl 4-hydroxylase that binds the proline-rich peptide are described. The crystals obtained diffract to a resolution of better than 3 Å.
Crystals of imidase from pig liver have been crystallized in the triclinic P1 space group and the orthorhombic C2221 space group.
The crystallization and preliminary characterization of unsaturated glucuronyl hydrolase from Bacillus sp. GL1 are presented.
A recombinant yeast cytosine deaminase has been crystallized, showing diffraction beyond 1.5 Å resolution, and an initial atomic model has been built.
D-Phenylglycine aminotransferase from P. stutzeri has been crystallized in the trigonal space group P3121 or P3221. A 2.3 Å resolution diffraction data set was collected.
short communications
The crystal structure of the single-chain Fv fragment of the monoclonal antibody 1696, which inhibits the protease of HIV-1 and HIV-2, has been determined in the non-complexed state at 1.7 Å resolution. It is compared with the previously determined structures of the non-complexed Fab1696 and scFv1696 as an antibody–antigen complex.
PDB reference: Fv fragment of HIV protease inhibitor, 1n4x, r1n4xsf
book reviews
Free 



 journal menu
journal menu