issue contents
January 2004 issue
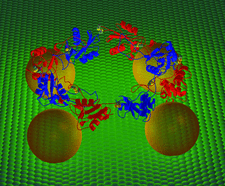
Cover illustration: Schematic representation of Convulxin tetramer interaction with protein ligands at the platelet cell surface (p. 46).
10th anniversary papers
The first structure of an L-RNA duplex is compared with its D-RNA analogue.
PDB reference: RNA in L-configuration, 1r3o, r1r3osf
The structure of the RNA dodecamer demonstrates the stability of the mismatched base pairing conformation of UUCG in RNA double-helical structures in the crystalline state.
NDB reference: r(GAUCACUUCGGU), AR0007
research papers
The 1.55 Å resolution three-dimensional structure of a squash glycerol-3-phosphate acyltransferase (GPAT) was determined using the multiwavelength anomalous dispersion method. This structure analysis and chimeric studies of plant GPATs will enable us to elucidate the mechanism involved at the atomic level in a plant's tolerance of chilling temperature.
PDB reference: glycerol-3-phosphate acyltransferase, 1iuq, r1iuqsf
The complex multivariate normal distribution is applied in a maximum-likelihood formalism to the single-wavelength anomalous diffraction experiment, producing better results compared with currently used programs in the test cases shown.
The magnitude of the anomalous signal of a xenon complex of porcine pancreatic elastase has been investigated as a function of data-collection wavelength and scaling protocol.
PDB reference: porcine pancreatic elastase–Xe complex, 1uo6, r1uo6sf
The usefulness of flash-cooling in a capillary (FCC) is described. The FCC method cools crystals more slowly than conventional cooling and may be applied when conventional cooling using a cryoloop fails.
The crystal structure of the toxin convulxin from the venom of the South American rattlesnake C. durissus terrificus has been solved by molecular replacement and refined at 2.7 Å resolution. Analysis of the structure provides insights into the interactions between convulxin and platelet receptors.
PDB reference: convulxin, 1uos, r1uossf
The structure of human Rab5a GTPase domain was refined at 1.05 Å resolution using the program SHELXL.
PDB reference: Rab5a GTPase domain, 1r2q, r1r2qsf
The crystal structures of two DNA octamers with different positions of double methylated cytosines reinforce the idea that methylation of cytosine stabilizes the A-type conformation for alternating DNA sequences beginning with a 5′-purine.
The crystal structure of an acidic phospholipase A2 from Indian saw-scaled viper (E. carinatus) has been determined at 2.6 Å resolution. It reveals an unusual intermolecular interaction involving the β-wing of one molecule and the calcium-binding loop of a symmetry-related molecule.
PDB reference: phospholipase A2, 1oz6
A general formula for phasing reflections via SAD/MAD data is provided; a procedure is described and successfully applied to various practical cases.
Thaumatin packing contacts in four crystal lattices are described.
PDB reference: thaumatin, 1pp3, r1pp3sf
The crystal structure of a DNA fragment d(GCGAAAGC) shows that two octamers related by a crystallographic twofold symmetry are associated to form a base-intercalated duplex, which is maintained by two Watson-Crick G·C base pairs and a subsequent sheared G·A pair at both ends, the central four A residues being stacked on each other and exposed suggesting a functional role.
The crystal structure of phosphopantetheine adenylyltransferasefrom T. thermophilus HB8 complexed with Ppant at 1.50 Å resolution is reported.
PDB reference: 1od6, r1od6sf
crystallization papers
The effector-binding domains of two LysR-type transcriptional regulators, BenM and CatM, were crystallized. Additionally, the effector-binding domain of BenM was crystallized in the presence of effectors.
Two fragments of the C-terminal catalytic domain of human poly(ADP-ribose) polymerase (catPARP), Met-catPARP and Gly Ser-catPARP, were purified and crystallized. The crystals show diffraction to at least 3.5 Å resolution.
Three crystal forms of the NAC domain of the A. thaliana ANAC protein have been obtained. One of the crystal forms is suitable for structure determination, which is being pursued by multiple isomorphous replacement.
Recombinant B. subtilis and A. thaliana MTR kinases were expressed, purified and crystallized. A colorimetric enzyme-activity assay and preliminary X-ray analysis of the crystals are described.
The primary dehydratase of the fatty-acid elongation cycle, FabZ (β-hydroxyacyl ACP dehydratase), from P. falciparum has been crystallized.
L7Ae sRNP core protein from P. abyssi was crystallized and data were collected to 2.9 Å resolution.
The crystallization and preliminary X-ray study of the CTX-M-type extended-spectrum β-lactamase SED-1 from C. sedlakii and its mutant SED-G238C are reported.
The 21 kDa N-terminal domain of XpsE protein from X. campestris has been crystallized in the tetragonal space group P41212 (or P43212), with unit-cell parameters a = b = 56.1, c = 102.7 Å. A complete native data set to 2.0 Å has been collected.
To explore the molecular origin of the conformational stabilization mechanism of αTS, the α-subunit protein has been overexpressed in E. coli and crystallized using the hanging-drop vapour-diffusion method at 298 K.
A fragment of the rotaviral enterotoxin NSP4 containing the viral virulence-determining peptide preceded by the diarrhoea-inducing coiled coil region has been crystallized. X-ray diffraction data have been collected to a resolution of 2.2 Å.
Co-crystallization of peptide deformylase from Leptospira interrogans with its natural inhibitor actinonin produced diffraction-quality crystals that belong to space group P21, with unit-cell parameters a = 87.5, b = 119.1, c = 95.8 Å, β = 111.6°.
The conserved ribonucleoprotein core of the signal recognition particle (SRP) has been crystallized. Both crystal forms are highly twinned and an explanation for the possible tetartohedral twinning is presented.
An antitumour pepleomycin:d(CCAGGCCTGG) complex has been crystallized. The crystals belong to space group P212121 and diffraction data have been collected to 2.8 Å resolution.
The quorum-sensing regulator TraM from A. tumefaciens has been crystallized and crystals diffract to 1.67 Å using synchrotron radiation. Selenomethionine-substituted TraM has also been crystallized and a two-wavelength MAD data set has been collected to 2.0 Å resolution.
X-ray crystallography of a unique dye-decolorizing peroxidase is reported.
The crystallization and preliminary X-ray diffraction analysis of the catalytic subunit of A. thaliana AHAS in complex with the sulfonylurea herbicide chlorimuron ethyl are reported.
Crystals of Nc-rck/p54 have been grown and diffract to beyond 2 Å resolution.
To understand the structural basis of thermostability, three crystal forms of B. subtilis lipase have been obtained at low pH along with three crystal forms of thermostable mutants generated by directed evolution.
Two crystal forms and the preliminary X-ray data of the T. reesei hydrophobin HFBII are described.
YsxC, a putative GTP-binding protein from B. subtilis, has been crystallized in the presence and absence of GDP in forms suitable for structure determination.
Three different diffraction-quality crystals of the catalytic domain of PDE3B have been obtained by the vapor-diffusion method. Structure solution is under way.
Fab fragments of a mouse monoclonal carbamatase catalytic antibody have been prepared and crystallized in complex with a transition-state analogue in a form suitable for structure determination.
AppA, a receptor protein for oligopeptide uptake by B. subtilis, has been crystallized and native diffraction data to 2.3 Å spacing have been collected using synchrotron radiation.
Geranylgeranyl diphosphate synthase and its SeMet derivative have been expressed in E. coli and crystallized in space group P41 or P43. Diffraction data from both crystals have been collected using the SPring-8 synchrotron-radiation source.
Crystals of the G. diazotrophicus levansucrase have been obtained and diffract to 2.5 Å spacing using a synchrotron X-ray source.
PhzF, an enzyme that catalyzes a key condensation reaction in the biosynthesis of phenazine-1-carboxylic acid in P. fluorescens, was cloned, overexpressed and crystallized. Data collection from native and seleno-L-methionine-labelled crystals is reported.
The proteins Spe31 and Spe32 were purified simultaneously from the seeds of P. erosus. Crystals were obtained from the protein mixture by the hanging-drop vapour-diffusion method and they diffracted to a resolution of 2.61 Å on an in-house X-ray source. Gel electrophoresis under non-denaturing conditions showed that the protein crystallized was Spe31.
Crystals of pectate lyase from E. chrysanthemi have been obtained. Diffraction data have been collected from a native crystal and a gold derivative.
Crystals of pyruvate phosphate dikinase from maize have been obtained by the vapour-diffusion method. The crystals diffract to 2.3 Å using synchrotron radiation and belong to space group C2, with unit-cell parameters a = 108.2, b = 100.2, c = 108.4 Å, β = 96.5°.
The crystallization of recombinant phosphopantetheine adenylyltransferase (PPAT), the penultimate enzyme in coenzyme A (CoA) biosynthesis, from M. tuberculosis is reported.
Crystals of T. multicolor P2Ox have been grown from monomethylether PEG 2000, sodium acetate, MgCl2 and Ta6Br12. They belonged to space group P21, with unit-cell parameters a = 99.9, b = 101.7, c = 135.6 Å, β = 90.85°.
short communications
W3Y single mutant of rubredoxin from Pyrococcus furiosus: a preliminary time-of-flight neutron study
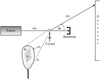
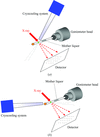
Rubredoxin from the hyperthermophilic archaeon P. furiosus maintains its native structure at high temperatures (373 K). In order to investigate the role of hydrogen bonding, hydration and chain dynamics in this thermostability, wavelength-resolved Laue neutron diffraction data have been collected from the W3Y single mutant (Trp3→Tyr3) on the spallation neutron protein crystallography station (PCS) at Los Alamos Neutron Science Center.
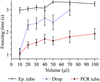
This paper describes a method for long-term storage of proteins for crystallography by freezing samples in PCR tubes. The use of PCR tubes results in more rapid freezing than the more traditional technique of dropping protein directly into liquid nitrogen and can successfully preserve samples that are damaged by the drop-freezing technique.
international union of crystallography
Free 



 journal menu
journal menu