issue contents
November 2004 issue
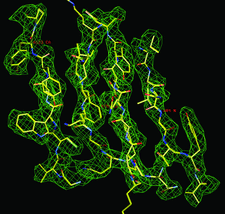
Cover illustration: Electron-density map of the protein histone methyltransferase SET 7/9 contoured at 1![[sigma]](/logos/entities/sigma_rmgif.gif) calculated from the peak-wavelength Se-SAD data. Phases were derived by OASIS-2004 and improved by DM. The data were provided by Drs S. J. Gamblin and B. Xiao. The protein was originally determined by J. R. Wilson et al. [(2002), Cell, 111, 105-115] using the MAD method. The refined structure model from the original authors is superimposed on the electron-density map (p. 1987).
calculated from the peak-wavelength Se-SAD data. Phases were derived by OASIS-2004 and improved by DM. The data were provided by Drs S. J. Gamblin and B. Xiao. The protein was originally determined by J. R. Wilson et al. [(2002), Cell, 111, 105-115] using the MAD method. The refined structure model from the original authors is superimposed on the electron-density map (p. 1987).
research papers
The structure of human NADH-cytochrome b5 reductase at 1.75 Å resolution is the same as that of rat NADH-cytochrome b5 reductase except for a large main-chain shift caused by a Pro268Thr substitution. Five mutations seem to be related to cytochrome b5.
PDB reference: NADH-cytochrome b5 reductase, 1umk, r1umksf
Open  access
access
 access
accessPeptidic molecules in the CSD are searched for nests, three-amino-acid residue anion-binding motifs found commonly in proteins. 37 are found. One, in the antibiotic vancomycin, has the function of binding the C-terminal carboxylate group of the bacterial cell wall precursor peptide.
In the course of investigating the complex of HIV-1 protease with a novel phenylnorstatine inhibitor, two inhibitor molecules were found to be bound to the enzyme, one in a non-productive mode. The contribution of the contacts of this inhibitor molecule at the protein interface to the crystal quality is discussed.
The structure of 2C-methyl-D-erythritol-2,4-cyclophosphate from S. oneidensis MR1 (SO3437) has been determined to 1.6 Å resolution in the absence of substrate.
The effects of radiation damage on iodinated thaumatin crystals have been analysed. It was shown that combination of the anomalous and isomorphous signals in the form of RIPAS (radiation-damage-induced phasing with anomalous scattering) is beneficial for both locating the substructure and subsequent phasing.
The crystal structure of E. coli argininosuccinate lyase has been determined at 2.44 Å resolution; in this structure, one active site, complexed with two phosphate ions, adopts the closed conformation.
PDB reference: argininosuccinate lyase, 1tj7, r1tj7sf
Download citation


Download citation


The D,L-alternating peptide H-(L-Tyr-D-Tyr)4-L-Lys-OH adopts a gramicidin A-like structure. Fragment-search strategies aiming to exploit the high-resolution data available (1.3 Å) were tested.
PDB reference: H-(L-Tyr-D-Tyr)4-L-Lys-OH, 1uno, r1unosf
De novo crystallization of a ternary complex composed of Y. pestis virulence factors YopN, SycN, and YscB could only be achieved after reductive methylation of surface-exposed lysine residues. This case study also demonstrates that the modification can be safely employed on multiprotein complexes and selenomethionine-substituted samples, underscoring its general potential as a crystallization tool, even in high-throughput settings.
The influence of the error term on direct-method SAD phasing is examined. A method for automatically tuning the scaling factor in the error term is proposed.
Structure fragments resulting from automatic model building based on initial direct-method SAD phasing are fed back to the direct-method probability formula in order to strengthen the phasing power and to benefit the automation of structure solution of proteins.
structural genomics papers
The X-ray crystallographic structure of T. thermophilis RraA has been solved to 2.3 Å resolution.
PDB reference: RraA, 1j3l, r1j3lsf
crystallization papers
The last step of the shikimate pathway is catalyzed by chorismate synthase (CS). Optimization of crystallization trials allowed the crystallization of homogeneous recombinant CS from M. tuberculosis.
A thermostable homing endonuclease from Thermoproteus sp. IC-061 has been crystallized. The crystals diffract to 2.7 Å resolution.
Open  access
access
 access
accessThe full-length single-stranded DNA-binding protein (SSB) from the extremophile A. aeolicus has been expressed in Escherichia coli, purified and crystallized and initial X-ray data have been obtained.
Open  access
access
 access
accessPreliminary crystallographic data of the fusion core of the spike protein of the murine coronavirus mouse hepatitis virus have been obtained.
Crystals of ChiF1 have been grown and they diffract beyond 1.24 Å resolution.
Human liver regucalcin, consisting of 299 amino-acid residues, has been overexpressed in Escherichia coli, purified and crystallized by the vapour-diffusion method in the presence of polyethylene glycol 4000 as a precipitant.
VCBP3, an immunoglobulin-like protein from the protochordate B. floridae with the characteristics of a `primordial' antigen receptor, has been crystallized and complete native data sets have been collected to 2.4 Å resolution. Isomorphous crystals from two different crystallization conditions are consistent with space groups P3121 and P3221, with unit-cell parameters a = b = 58.99, c = 79.21 Å, α = β = 90, γ = 120°.
The expression, purification, crystallization and preliminary X-ray diffraction studies of the protein TraF from the E. coli F plasmid are reported.
Vacuolar sorting receptor (VSR) proteins target ligands to the lytic vacuole in a sequence-specific manner. Purification from Drospohila S2 cells, crystallization and preliminary X-ray diffraction studies of Arabidopsis thalia VSR, AtBP80b, have been carried out.
Open  access
access
 access
accessGlutamate racemase from B. subtilis has been crystallized in the presence of L-glutamate in a form suitable for structure determination.
Aall-A and Aall-B, two novel heterodimeric snake-venom C-type lectin-like proteins (sv-CLPs), were purified from the venom of D. acutus from Anhui, China, and crystallized.
The M. oreades agglutinin (MOA) has been crystallized. Crystals diffract anisotropically to 2.5 Å resolution. A complete X-ray data set has been obtained to 3.0 Å resolution.
The ectoplasmic region of apical membrane antigen 1 from P. vivax has been expressed and crystallized in two crystal forms. Phases have been obtained by the MAD technique using a Pt derivative.
The water-forming NADH oxidase from L. sanfranciscensis has been crystallized in space group P212121 and X-ray diffraction data to 1.85 Å resolution have been collected.
The retinoic acid receptor β ligand-binding domain has been expressed and crystallized bound to a synthetic retinoid (TTNPB) with or without a co-activator peptide (SRC-1).
Crystals of infestin 4, a factor XIIa Kazal-type inhibitor from T. infestans, diffracted to 1.8 Å resolution and belong to the orthorhombic space group P212121, with unit-cell parameters a = 25.89, b = 45.64, c = 57.41 Å.
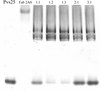
Crystals of the complex between the P. vivax sexual stage 25 kDa protein Pvs25 and a malaria transmission-blocking Fab diffract X-rays to 3.5 Å. Two complex molecules are predicted per asymmetric unit.
Mexicain, a cysteine protease, has been extracted from the latex of the tropical plant Pileusmexicanus, purified and crystallized. The crystals belong to the space group P21 and they diffract X-rays to 2.1 Å resolution.
Pyridoxal 4-dehydrogenase from Microbacterium luteolum has been crystallized by the sitting-drop vapour-diffusion method. The crystals were monoclinic and belonged to space group C2, with unit-cell parameters a = 107.0, b = 56.7, c = 130.2 Å, β = 103.6°. Diffraction data were collected from a single crystal to 2.0 Å.
Open  access
access
 access
accessThe catalytic domain of one of three sialidases encoded by the C. perfringens genome has been subcloned and crystallized. The diffraction of this 50 kDa domain to beyond 0.92 Å will allow detailed mechanistic enzymology of this enzyme superfamily.
The crystallization of spermidine synthase from H. pylori is reported.
Sulfite dehydrogenase from S. novella has been crystallized. Crystals diffract to at least 1.8 Å resolution and belong to space group P21212, with unit-cell parameters a = 97.5, b = 92.5, c = 55.9 Å.
Crystals of wild-type and two mutants of apo tryptophanase from E. coli belong to space group F222 and diffract to 1.9 Å.
A protein disulfide oxidoreductase from A. aeolicus has been crystallized for the first time. Crystals belong to space group R32 and diffract to 2.4 Å resolution.
The hook is a short, highly curved segment of the bacterial flagellum, working as a universal joint, and is a helical assembly of about 120 copies of a single protein FlgE (42 kDa). The expression, purification, crystallization and diffraction data collection of a 31KDa fragment of FlgE, which is otherwise extremely difficult to crystallize because of its strong polymerization ability, are reported.
Chitosanase from Bacillus sp. strain K17, which belongs to glycoside hydrolase family 8 and exhibits subclass II specificity, has been crystallized in two forms corresponding to the active and inactive states.
Mouse autocrine motility factor has been crystallized in the absence and presence of carbohydrate phosphate inhibitors. Diffraction data have been collected using synchrotron radiation.
A stable domain of the vesicular stomatitis virus phosphoprotein was crystallized. The crystals belonged to space group P41212 or P43212, with unit-cell parameters a = b = 74.50, c = 156.84 Å, and diffraction data were collected to 2.75 Å.
The production and crystallization of recombinant E. coli glutamate-5-kinase, the enzyme that catalyzes the first and feedback-controlled step of proline biosynthesis and a member of the amino-acid kinase enzyme family, is reported. In addition to the catalytic machinery, this enzyme contains a PUA putative RNA-binding domain that endows it with gene-expression regulatory properties.
Crystals of the catalytic fragment of human 2′,3′-cyclic nucleotide 3′-phosphodiesterase (hCNP-CF) have been obtained. The crystals diffract X-rays to 1.8 Å resolution.
PEX1 N-terminal domain crystals have been prepared. The crystals belong to space group P31 or P32, with unit-cell parameters a = b = 63.5, c = 33.5 Å, and contain one protein molecule per crystallographic asymmetric unit. An intensity data set was collected to a resolution of 2.05 Å.
Purification, identification and preliminary crystallographic studies on an allergy-related protein from V. unguiculata seeds were carried out. The protein, crystals of which diffract to about 2.1 Å resolution, does not indicate homology with any protein with known crystal structure.
The banana lectin from M. paradisiaca has been purified and crystallized and the crystal structure studied.
A proteolyzed form of horse PLRP2 has been crystallized by the hanging-drop vapour-diffusion method in complex with DDAO. Native data have been collected to 2.9 Å resolution.
short communications
Free 

This work describes the structure of PhzG from two species of Pseudomonas.
addenda and errata
Free 



 journal menu
journal menu