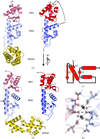issue contents
October 2007 issue
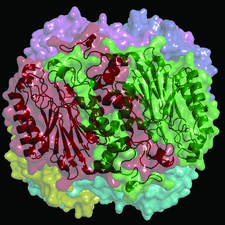
Cover illustration: A view down the twofold axis of the hexameric amidase (AmiE) from Bacillus pallidus RAPc8 which is a member of the nitrilase superfamily of enzymes. (p. 1048).
research papers
The scalar approximation to structure-factor extrapolation (SASFE) procedure is used to extrapolate structure-factor amplitudes for a fully `activated' crystal from amplitudes of a native and a partially activated crystal. Here, it is investigated whether and under what conditions the approximations inherent in SASFE introduce significant errors. The results indicate that the errors introduced by SASFE's assumptions are negligible and that SASFE's usefulness in macromolecular crystallography is limited primarily by the propagation of experimental uncertainties.
The crystal structure of St-AspRS (nondiscriminating type) has been determined at 2.3 Å resolution as the first example of the nondiscriminating type of AspRS from crenarchaea.
PDB reference: aspartyl-tRNA synthetase, 1wyd, r1wydsf
The homohexameric structure of a member of the nitrilase superfamily with D3 symmetry. The active site configuration suggests a catalytic role for Glu142.
PDB reference: aliphatic amidase, 2plq, r2plqsf
The crystal structure of P. horikoshii TYW1 is described.
PDB reference: TYW1, 2yx0, r2yx0sf
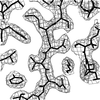
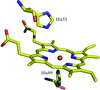
The crystal structure of NovP, an O-methyltransferase comprising 262 amino acids, was solved by SIRAS using 1.35 Å native data and 2.45 Å data from a single mercury derivative using a combination of partial chain tracing by hand of the initial very noisy map followed by data extrapolation to 1.0 Å resolution.
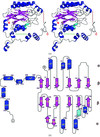
The first two winged-helix motifs of domain IV of translation elongation factor SelB from M. thermoacetica can undergo conformational switches, which could support a functional interaction between the N- and C-terminal portions of SelB.
PDB reference: WH1 and WH2 motifs of SelB, 2v9v, r2v9vsf
The crystal structure of the red fluorescent protein zRFP574 from Zoanthus sp. 2 at 1.51 Å resolution allowed a detailed description of the chromophore derived from decarboxylation of the aspartate side chain within the chromophore-forming tripeptide Asp-Tyr-Gly. Comparison with the newly determined X-ray structure of the green fluorescent protein zGFP506 from a related Zoanthus species and its mutant zGFP506_N66D in the transitional `green' and mature `red' forms provides new insight into the structural chemistry responsible for the acquisition of red fluorescence.
In order to investigate the dual function of the dehaloperoxidase from the terebellid polychaete Amphitrite ornata, the enzyme was expressed in Escherichia coli as a recombinant protein in its wild-type form and as a mutant protein. Both the wild-type and mutant proteins were crystallized and their structures were determined at 100 K to a resolution of 1.62 Å.
Sylvaticin, a 10 kDa protein excreted by the Oomycete Pythium sylvaticum reveals an α-elicitin-like structure with a reduced internal cavity.
short communications
The crystal structure of RbcX from Synechocystis sp. PCC6803 is reported, revealing an unusually long protruding C-terminal helix, as well as a bound polyethylene glycol molecule in the protein substrate-binding site.
PDB reference: RbcX, 2py8, r2py8sf
letters to the editor
Free 



 journal menu
journal menu