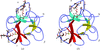issue contents
January 2009 issue
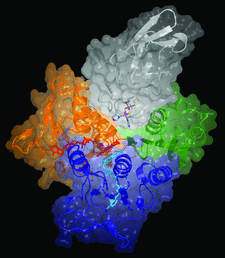
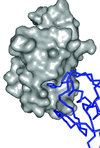
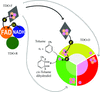
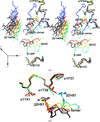
Cover illustration: The three-component Rieske non-heme iron toluene 2,3-dioxygenase system catalyses the hydroxylation of many aromatic hydrocarbon substrates (p. 24). The system consists of a reductase, ferredoxin, and a terminal dioxygenase. The cover figure illustrates a modelled binary complex between the three-domain reductase (blue, green, orange) and ferredoxin (grey) during the initial electron transfer. Two electrons are transferred from NADH (cyan) in the hydride transfer to FAD (red) in the reductase. The ferredoxin is believed to dock between the FAD-binding domain (orange) and C-terminal domain (green) to shuttle one electron from the FAD to the active site of the dioxygenase.
research papers
An analysis of the different patterns of subunit association observed in the crystal structures of various homotetrameric proteins is presented.
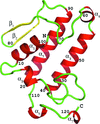
The structure of the effector domain of the influenza protein NS1, a validated antiviral drug target, has been solved in two space groups.
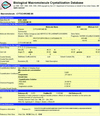
Improvements to the Biomolecular Crystallization Database are described, including new search features and expanded content. A few examples of searches and analysis are given.
The crystal structures of the three-component toluene 2,3-dioxygenase system provide a model for electron transfer among bacterial Rieske non-heme iron dioxygenases.
PDB references: TDO-R, 3ef6, r3ef6sf; TDO-F, 3dqy, r3dqysf; TDO-O, 3en1, r3en1sf; apo-TDO-O, 3eqq, r3eqqsf
The enzyme dehaloperoxidase (DHP) from the terebellid polychaete Amphitrite ornata, which can function as both a hemoglobin and a peroxidase, has been studied.
PDB reference: deoxy dehaloperoxidase, 3dr9, r3dr9sf
The crystal structure of a human carbonmonoxy hemoglobin has been determined at 2.8 Å resolution. The structure, which is in a relaxed conformation, shows rotation of βHis63(E7) out of the distal pocket, creating a ligand channel to the solvent.
PDB reference: relaxed-state human hemoglobin, 3d17, r3d17sf
Crystal structures of the C-half domain of the galactose-binding lectin EW29 from the earthworm L. terrestris in complex with D-lactose and N-acetyl-D-galactosamine were determined.
The X-ray crystal structures of the BIR3 domain of cIAP1 in complex with N-terminal peptides from SMAC and caspase-9 have been determined. The binding affinities of the complexes were determined using the surface plasmon resonance technique. The structures were compared with similar structures of XIAP.
Structural analysis of a highly stable FGF-1 mutant based on 1.70 Å crystal structure reveals three localized conformational changes.
The water molecules in crystal structures of bovine pancreatic phospholipase A2 have been investigated in order to identify the invariant water molecules and their possible roles in the structure and function of the enzyme.
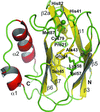
The atomic resolution structure of pseudoazurin from Hyphomicrobium denitrificans demonstrates that its spectroscopic properties are a consequence of its characteristic Cu coordination geometry.
PDB reference: pseudoazurin, 3ef4, r3ef4sf
obituaries
Free 

international union of crystallography
Free 



 journal menu
journal menu