issue contents
March 2009 issue
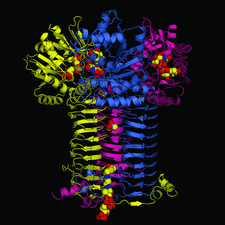
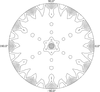
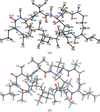
Cover illustration: GlmU is the bifunctional acetyltransferase/uridyltransferase that catalyses the conversion of GlcN-1-P to UDP-GlcNAc, a key substrate in lipopolysaccharide and peptidoglycan synthesis. The cover figure shows the biological trimer of the M. tuberculosis enzyme, an important drug target, complexed with the product UDP-GlcNAc (top: uridyltransferase active site) and with cacodylate anion (middle: on the crystallographic threefold axis). The acetyl-coA cofactor has been modelled in the acetyltransferase active site (bottom) (p. 275).
research papers
Open  access
access
 access
accessThe structures of the two alternatively spliced isoforms of human ketohexokinase, hepatic KHK-C and peripheral KHK-A, and of the ternary complex of KHK-A with the substrate fructose and AMP-PNP have been solved. The differences between KHK-A and KHK-C resulting from the spliced region are subtle and affect thermostability and probably flexibility; the mutations causing fructosuria were modelled.
The structure of the DUF55 domain of human thymocyte nuclear protein 1, which was determined from partially tetartohedrally twinned crystals, is reported.
The structure of the lysosomal 66.3 kDa protein, which crystallizes in the monoclinic space group C2, was solved by means of sulfur SAD phasing using data with an Ranom/Rp.i.m. ratio of 1.1.
PDB reference: lysosomal 66.3 kDa protein, 3fbx, r3fbxsf
The structure of SoxS has been solved in oxidized and reduced forms at 2.1 and 1.9 Å resolution, respectively.
Open  access
access
 access
accessThe crystal structures of DAPK–ADP–Mg2+ and DAPK–AMP-PNP–Mg2+ complexes were determined at 1.85 and 2.00 Å resolution, respectively. Comparison of the two nucleotide-bound states with apo DAPK revealed localized changes in the glycine-rich loop region that were indicative of a transition from a more open state to a more closed state on binding of the nucleotide substrate and to an intermediate state with the bound nucleotide product.
A revised EDM–DEDM algorithm for phase refinement is described, together with its application to proteins, with special attention being given to molecular-replacement cases.
Structure of the G225P/G226P mutant of mouse 3(17)α-hydroxysteroid dehydrogenase (AKR1C21) ternary complex is reported at 2.1 Å resolution.
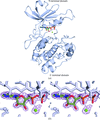
The crystal structure of C2b has been determined at 1.8 Å resolution, which reveals the arrangement of its three complement control protein (CCP) modules. A model for complement component C2 is presented and its conformational changes during the C3-convertase formation are also discussed.
PDB reference: C2b, 3erb, r3erbsf
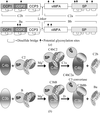
The crystal structure of M. tuberculosis GlmU has been determined in an unliganded form and in complex with UDP-GlcNAc or GlcN-1-P. NMR-based enzymatic activity assays suggest that the presence of acetyl-coenzyme A has an inhibitory effect on uridyltransferase activity.
Download citation


Download citation


Two single-crystal X-ray diffraction data sets for cyclosporine A were measured to high resolution using synchrotron radiation at temperatures of 5 and 90 K. They allowed an accurate determination of its molecular and electronic structure.
short communications
The structure of uncomplexed plasmepsin II from P. falciparum is shown to contain noncoplanar catalytic aspartic acid residues.
PDB reference: plasmepsin II, 3f9q, r3f9qsf
Open  access
access
 access
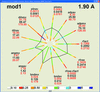
accessThe representation of crystallographic model characteristics in the form of a polygon allows the quick comparison of a model with a set of previously solved structures.
addenda and errata
Open  access
access
 access
access

 journal menu
journal menu