issue contents
November 2009 issue
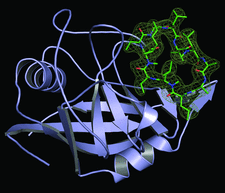
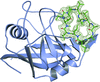
Cover illustration: Crystal structure of cyclophilin from L. donovani complexed with cyclosporin at 2.6 Å resolution (p. 1187). The electron density around cyclosporin is contoured at the 2.0![[sigma]](/logos/entities/sigma_rmgif.gif) level from a 2Fo - Fc map.
level from a 2Fo - Fc map.
research papers
The nucleation of lysozyme in microbatch experiments was linked to the formation of protein–precipitant interfaces. The use of oscillatory shear allowed decreasing the nucleation rate and extending the growth period for lysozyme crystals, presumably through the control of the number of interfaces and removal of impurities or defects.
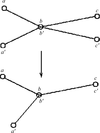
Criteria for the interpretability of coordinate differences and a new method for identifying rigid-body motions and nonrigid deformations in protein conformational changes are developed and applied to functionally induced and crystallization-induced conformational changes.
Here, the techniques, tactics and strategies used to overcome a series of technical roadblocks in crystallization and phasing of the trimeric ebolavirus glycoprotein are described.
The structure of the nucleotide-binding domain of the P-type ATPase MgtA was determined and refined to 1.6 Å resolution.
PDB reference: Mg-ATPase nucleotide-binding domain, 3gwi, r3gwisf
The crystal structure of cyclophilin from Leishmania donovani complexed with cyclosporin A has been solved at 2.60 Å and the thermodynamics of binding has been correlated with the structure.
PDB reference: cyclophilin–cyclosporin A complex, 3eov, r3eovsf
The crystal structure of the fungal aryl alcohol-oxidase from Pleurotus eryngii, which catalyzes dehydrogenation reactions on different primary polyunsaturated alcohols yielding the corresponding aldehydes, has been determined.
PDB reference: aryl-alcohol oxidase, 3fim, r3fimsf
The crystal structure of Dau c 1, the major carrot allergen has been determined to a resolution of 2.7 Å. This enables insight into the molecular basis of cross reactivity between pollen and related food allergens.
PDB reference: Dau c 1, 2wql, r2wqlsf
During the improvement of initial direct-method SAD phases, combining the resultant phases in each stage with the original unresolved bimodal phase distributions from the SAD experiment is equivalent to rebreaking the phase ambiguity using a set of ever-improving phases. This significantly enhances the power of direct-method SAD phasing.
The 3.2 Å resolution crystal structure of the filamin A actin-binding domain reveals the localization of mutated residues in patients suffering from periventricular nodular heterotopia or otopalatodigital syndrome spectrum disorders.
PDB reference: human filamin A actin-binding domain, 2wfn, r2wfnsf
The importance of active-site electrostatics for oxidative and reductive half-reactions in a redox flavoenzyme (cholesterol oxidase) have been investigated by a combination of biochemistry and atomic resolution crystallography. A detailed examination of active-site dynamics demonstrates that the oxidation of substrate and the re-oxidation of the flavin cofactor by molecular oxygen are linked by a single active-site asparagine.
short communications
Simple and inexpensive methods have been developed so that phytochromes and other photochromic molecules can effectively be handled and crystallized. These have been demonstrated by the solution of the three-dimensional structure of the cyanobacterial Cph1 sensory module.


 journal menu
journal menu