issue contents
August 2010 issue
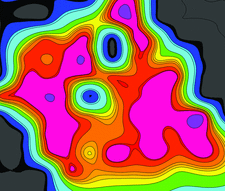
Cover illustration: A diffraction quality map of a crystal of bovine mitochondrial F1-ATPase as probed by a microfocused X-ray beam (p. 855). The map is coloured by the ranking resolution from EDNA (blue to mauve), contour lines mark areas of similar diffraction quality. The technique of diffraction cartography defines both the shape and internal order of crystals.
research papers
New tools for the evaluation of the diffraction characteristics of crystals of macromolecules are presented in the context of the ESRF Upgrade MASSIF beamlines.
The medium-resolution structure of the N-terminal fragment of E. coli Lon protease shows that this part of the enzyme consists of two compact domains and a very long α-helix.
PDB reference: N-terminal fragment of Lon protease, 3ljc
The crystal structure of the nonstoichiometric complex of gramicidin D with NaI has been studied using synchrotron radiation at 100 K.
PDB reference: gramicidin D–NaI complex, 3l8l
Open  access
access
 access
accessThe 2.5 Å resolution structure of S. aureus adenylosuccinate lyase is reported and compared with those of orthologues to assess its potential as a template for early stage drug discovery. AMP and a putative assignment of oxalate, the latter an artefact possibly arising from an impurity in the PEG used for crystallization, occupy the active site.
PDB reference: adenylosuccinate lyase, 2x75
Guidelines and specific tests for validating macromolecular crystal structures that include TLS models are introduced. Validation may used to troubleshoot problems during refinement, to confirm the internal consistency of the model as part of deposition into the Protein Data Bank or to assess the plausibility of interpretating the boundary between two TLS groups as indicating a hinge point between structural domains.
A novel approach is presented for the visualization of crystallization experiments using fluorescence from trace amounts of a nonspecific dye.
The crystal structure of Rv0554 from M. tuberculosis has been determined, revealing a dimeric protein with an α/β-hydrolase fold. Functional assays failed to support a proposed role in menaquinone biosynthesis.
The molecular mechanism of the VDR-antagonistic action of TEI-9647 is investigated.
Open  access
access
 access
accessThe application of microseed matrix screening to the crystallization of related antibodies in complex with IL-13 is described. Both self-seeding or cross-seeding helped promote nucleation and increase the hit rate.
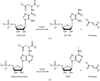
Crystallographic binding studies of a monomeric variant of triosephosphate isomerase (A-TIM) with a modified more extended binding pocket between loop-7 and loop-8 are reported. Wild-type TIM exclusively catalyses the isomerization of the C3-sugar phosphates dihydroxyacetonephosphate and D-glyceraldehyde-3-phosphate. These crystallographic studies show that the new binding pocket of A-TIM can also bind analogues of C4-sugar phosphates and C5-sugar phosphates.
A new statistical and computational procedure, which merges multiple FA estimates into an averaged data set, is used to further improve the quality of the estimated anomalous amplitudes for substructure determination.
short communications
The structure of human carbonic anhydrase II in the monoclinic P21 space group with a doubled a axis from that of the usually observed unit cell has been re-determined and shown that the choice for how the four molecules in the unit cell are grouped (based on their coordinates) into pairs that represent a single asymmetric unit determines whether or not rotational disorder is observed/created during refinement.
PDB reference: human carbonic anhydrase II, 3mwo


 journal menu
journal menu