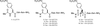issue contents
October 2010 issue
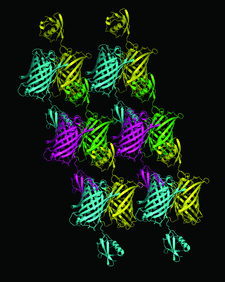
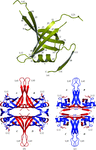
Cover illustration: A pair of fusion proteins forming a dimer through GFP-GFP interaction are coloured as magenta/green or cyan/yellow (p. 1059). The C-terminal ubiquitin moiety extends away from the N-terminal GFP moiety and interacts with the peripheral loops of the GFP moiety of another molecule.
research papers
The crystal structures of complexes of octahaem cytochrome c nitrite reductase from the bacterium T. nitratireducens (TvNiR) with sulfite and cyanide are reported. The sulfite reductase activity of TvNiR is quantitatively characterized.
Crystallographic and molecular-dynamics studies indicate a possible relation between variations in oligomerization and in stability, in addition to providing insights into changes in subunit structure on oligomerization.
The GFP-fusion technique has been successfully applied to the crystallization of small proteins and domains, using ubiquitin and the ubiquitin-binding motif as test cases.
C. perfringens α-toxin variant T74I has been shown to have significantly reduced cytotoxicity. Here, further characterization of the activity of this variant and its X-ray crystallographic structure are presented.
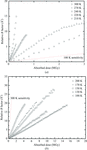
Overall and site-specific X-ray-induced damage to porcine pancreatic elastase was studied at atomic resolution at cryogenic temperatures of 100 and 15 K.
PDB references: porcine pancreatic elastase, model M1, 100 K, 3mnb; model M2, 100 K, 3mnc; model M3, 100 K, 3mns; model M4, 100 K, 3mnx; model M5, 100 K, 3mo3; model M6, 100 K, 3mo6; model M7, 100 K, 3mo9; model M8, 100 K, 3moc; model A1, 100 K, 3mty; model A2, 100 K, 3odf; model A3, 100 K, 3mu0; model A5, 100 K, 3mu1; model B1, 15 K, 3mu4; model B2, 15 K, 3odd; model B3, 15 K, 3mu5; model B5, 15 K, 3mu8
Radiation damage to protein crystals exhibits two regimes of temperature-activated behavior between T = 300 and 100 K, with a crossover at the protein glass transition near 200 K. These results have implications for mechanistic studies of proteins and for structure determination when cooling to T = 100 K creates excessive disorder.
Structures of the Grb2 SH2 domain complexed with a series of flexible and constrained replacements of the phosphotyrosine residue in tripeptides derived from Ac-pYXN (where X = V, I, E and Q) were compared to determine what, if any, structural differences arise as a result of ligand preorganization.
short communications
Transmission electron microscopy reveals a hexameric organization for the 2C protein of echovirus 30.


 journal menu
journal menu