issue contents
July 2011 issue
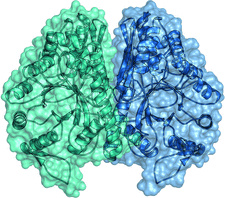
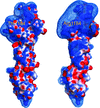
Cover illustration: Crystal structure of E. histolytica enolase (p. 619). Secondary-structure elements and a semi-transparent surface are shown in green for subunit A and in blue for subunit B.
research papers
This article presents the crystal structure of the T1184R variant of complement regulator factor H which has been associated with the development of atypical haemolytic uremic syndrome.
PDB reference: T1184R variant of complement regulator factor H, 3r62
The crystal structure of rabbit histidine triad nucleotide-binding protein 1 (rHINT1) in complex with adenosine is reported at 1.10 Å resolution.
PDB reference: rHINT1–adenosine complex, 3qgz
The development of methods to utilize X-ray diffraction data simultaneously collected from multiple single crystals is investigated. Data sets obtained from up to seven crystals of the proteins lysozyme and insulin showed good data quality.
The crystal structure of the glycolytic enzyme enolase from the pathogenic parasite E. histolytica is presented.
PDB reference: E. histolytica enolase, 3qtp
Data processing of an incommensurately modulated profilin–actin crystal is described.
The first X-ray crystal structure of the Y. pestis protein tyrosine phosphatase YopH in complex with an isothiazolidinone-based lead-fragment compound is reported.
An optically, UV and X-ray transparent crystallization plate suitable for in situ analysis has been developed. The plate uses contact line pinning rather than wells to confine the liquids.
Bacterial acyl carrier protein synthase plays an essential role in the synthesis of fatty acids, nonribosomal peptides and polyketides. In Mycobacterium tuberculosis, AcpS or group I phosphopentatheine transferase exhibits two different structural conformations depending upon the pH.


 journal menu
journal menu