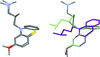issue contents
May 2016 issue
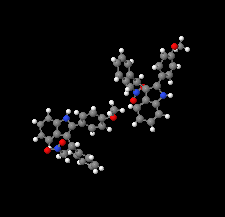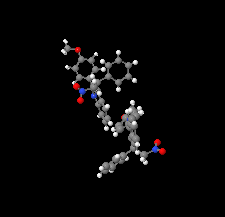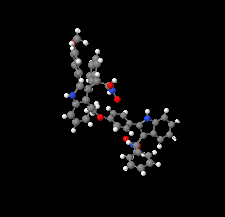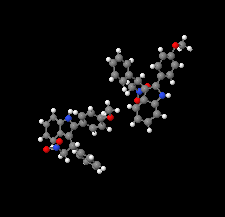
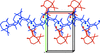
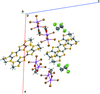
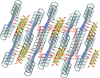
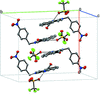
Cover illustration: N-H
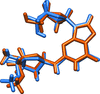
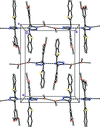
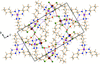
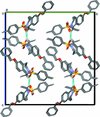
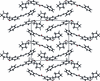
![[pi]](/logos/entities/pi_rmgif.gif) interactions are now a well-recognised type of `non-classical' weak bond and are of special significance in biological systems. For the indole derivatives 6-isopropyl-3-(2-nitro-1-phenylethyl)-1H-indole and 2-(4-methoxyphenyl)-3-(2-nitro-1-phenylethyl)-1H-indole, despite the presence of O atoms as potential acceptors for classical hydrogen bonds, the dominant intermolecular interaction in each crystal is an N-H
interactions are now a well-recognised type of `non-classical' weak bond and are of special significance in biological systems. For the indole derivatives 6-isopropyl-3-(2-nitro-1-phenylethyl)-1H-indole and 2-(4-methoxyphenyl)-3-(2-nitro-1-phenylethyl)-1H-indole, despite the presence of O atoms as potential acceptors for classical hydrogen bonds, the dominant intermolecular interaction in each crystal is an N-H
![[pi]](/logos/entities/pi_rmgif.gif) bond. This leads to chains in the former derivative and inversion dimers in the latter; C-H
bond. This leads to chains in the former derivative and inversion dimers in the latter; C-H
![[pi]](/logos/entities/pi_rmgif.gif) interactions appear to reinforce the N-H
interactions appear to reinforce the N-H
![[pi]](/logos/entities/pi_rmgif.gif) bonds in each case. See: Kerr, Trembleau, Storey, Wardell & Harrison [Acta Cryst. (2016). E72, 699-703].
bonds in each case. See: Kerr, Trembleau, Storey, Wardell & Harrison [Acta Cryst. (2016). E72, 699-703].
research communications


















































































 journal menu
journal menu