issue contents
August 2005 issue
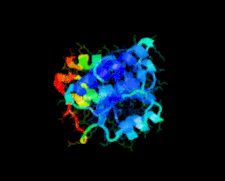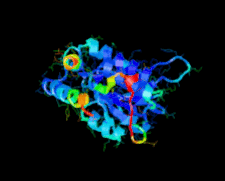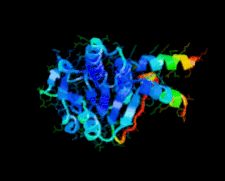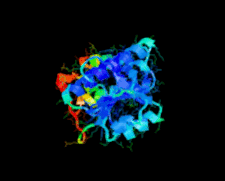
Cover illustration: A class II TrmH tRNA-modifying enzyme from Aquifex aeolicus (p. 722).
protein structure communications
The crystal structure of Aquifex aeolicus TrmH, a member of the a/b-knot superfamily responsible for O methylation of G18 of tRNAs, was determined to 1.85 Å resolution using the molecular-replacement method.
PDB reference: AaeTrmH, 1zjr, r1zjrsf
crystallization communications
Precipitation phase diagrams can be rapidly constructed using dispensing-robot technology. These diagrams provide information that assists in optimization of crystal growth.
Reaction centers from R. sphaeroides have been crystallized in a new crystal form in the presence of manganese.
Cloning, purification and crystallization of T. thermophilus proline dehydrogenase is reported. The detergent n-octyl β-D-glucopyranoside was used to reduce polydispersity, which enabled crystallization.
Adenosine 5′-monophosphate deaminase from A. thaliana has been crystallized in complex with coformycin 5′-phosphate. Diffraction data have been collected to 3.34 Å resolution.
Open  access
access
 access
accessSingle crystals of binary and ternary complexes of wild-type and D38C mutant H. mediterranei glucose dehydrogenase have been obtained by the hanging-drop vapour-diffusion method.
A family 10 alkali-thermostable xylanase from Bacillus sp. NG-27 has been crystallized. A diffraction data set has been collected to 2.2 Å resolution.
Crystals of pseudechetoxin and pseudecin, potent peptidic inhibitors of cyclic nucleotide-gated ion channels, have been prepared and X-ray diffraction data have been collected to 2.25 and 1.90 Å resolution, respectively.
A putative phosphoglycerate mutase from M. tuberculosis (Rv3214) has been crystallized. Diffraction data have been collected to 2.15 Å resolution from its selenomethionine-substituted form.
Open  access
access
 access
accessThis report describes the crystallization of a recombinant flavoprotein amine dehydrogenase/oxidase with specificity for L-proline from the hyperthermophile P. furiosus DSM 3638 and X-ray diffraction data collection. Crystals belonged to space group P1 and diffracted to a resolution of 3.3 Å.
This report describes the crystallization and X-ray diffraction data collection of three types (wild-type, W416F/V435W and C9S/C268S) of B. stearothermophilus. Crystals of C9S/C268S belonged to space group P6222 and diffracted to a resolution of 2.4 Å.
Open  access
access
 access
accessThe viral capsid protein P2 of bacteriophage PM2 has been crystallized. Preliminary X-ray analysis demonstrates the position and orientation of the two trimers in the asymmetric unit.
Crystals suitable for structural analysis have been prepared from a cell-wall invertase from A. thaliana.
Open  access
access
 access
accessThe detoxification enzyme glyoxalase I from L. major has been crystallized. Preliminary molecular-replacement calculations indicate the presence of three glyoxalase I dimers in the asymmetric unit.
Open  access
access
 access
accessCrystals of the complex formed between the bacterial membrane protein OmpC and the antibacterial protein lactoferrin suitable for high-resolution structure determination have been obtained. The crystals belong to the hexagonal space group P6, with unit-cell parameters a = b = 116.3, c = 152.4 Å.
Open  access
access
 access
accessImidazoleglycerol-phosphate dehydratase from A. thaliana has been overexpressed, purified and crystallized and data have been collected to 3 Å resolution.
Expression, purification and preliminary X-ray diffraction studies of the two domains of the sigma factor SigC from M. tuberculosis are reported.
M. tuberculosis diaminopimelate decarboxylase, the enzyme that catalyzes the final step of lysine biosynthesis, has been cloned, expressed, purified and crystallized in the absence of cofactor or substrate.
The hybrid moelcule of calmodulin and calmodulin-binding domain of olfactory nucleotide-gated ion-channel peptide (CaM-OLFp) was crystallized and preliminary analyzed using X-ray diffaction.
A 2:2 complex of highly purified GCSF receptor (Ig-CRH) with GCSF was crystallized. The crystal diffracted to 2.8 Å resolution with sufficient quality for further structure determination.
Crystals of the lipase of B. glumae in complex with its specific foldase were obtained in two forms. Crystallization, crystal manipulation and preliminary X-ray diffraction analysis are described.
The P. putida cytochrome P450cam operon repressor CamR has been expressed in E. coli and crystallized in space group P21212.
Δ1-Tetrahydrocannabinolic acid (THCA) synthase from C. sativa was crystallized. The crystal diffracted to 2.7 Å resolution with sufficient quality for further structure determination.
Open  access
access
 access
accessThe haem binding protein HemS from Y. enterocolitica has been crystallized in complex with its ligand. The crystals diffracted X-rays to 2.6 Å in-house.
A thaumatin-like antifungal protein, NP24-I, has been isolated from ripe tomato fruits. It was crystallized by the vapour-diffusion method and data were collected to 2.45 Å. The structure was solved by molecular replacement.


 journal menu
journal menu











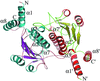






















![[publBio]](http://journals.iucr.org/logos/publbio.gif)





