issue contents
September 2005 issue
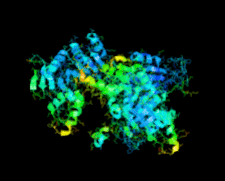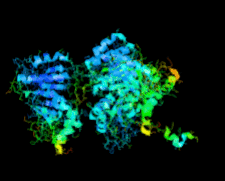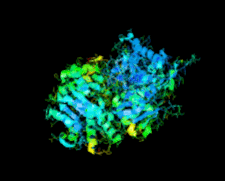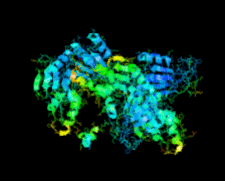
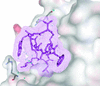
Cover illustration: Human basophilic leukemia-expressed protein BLES03 (p. 812).
protein structure communications
The crystal structure of porcine pancreatic elastase complexed with a novel trimacrocyclic peptide inhibitor FR901451 was solved at 1.9 Å resolution.
PDB reference: FR901451–PPE, 2cv3, r2cv3sf
structural genomics communications
The crystal structure of the 27.5 kDa BLES03 protein was determined at 2.5 Å resolution. Despite having an undetectable sequence relationship, the structure adopts a fold similar to that of eukaryotic initiation factor 4E with minor variations.
PDB reference: BLES03, 1ztp, r1ztpsf
This paper describes a Python script that may be used to gather all required structure-annotation information into an mmCIF file for upload through the RCSB PDB ADIT structure-deposition interface.
crystallization communications
Open  access
access
 access
access2,4-Dihydroxy-hepta-2-ene-1,7-dioate aldolase from E. coli C has been purified and crystallized. Diffraction data were collected to 1.6 Å and structure determination by molecular replacement is in progress.
Overexpression, purification and crystallization of C. metallidurans CopK allowed the collection of a complete data set to 2.2 Å resolution.
Overexpression, purification and crystallization of the YjgF/YER057c/UK114-family protein from S. tokodaii strain 7 allowed the collection of a complete data set to 2.0 Å resolution.
Human RGS10 and Gαi3 have been overexpressed and purified. Their complex has been crystallized (space group P43212 or P41212, unit-cell parameters a = 99.88, b = 99.88, c = 144.59 Å, α = β = γ = 90°) and diffraction data have been collected to 2.5 Å resolution using synchrotron radiation.
Crystallization of a family 84 glycoside hydrolase, a putative virulence factor, secreted by C. perfringens is reported.
Crystallization of the native and SeMet FMN reductase protein of the E. coli alkanesulfonate monooxygenase two-component enzyme system is reported.
Crystals of P. platycephala chintinase/lectin (PPL-2) belong to the orthorhombic space group P212121, with unit-cell parameters a = 55.19, b = 59.95, c = 76.60 Å. The preliminary cystal structure of PPL-2 was solved at a resolution of 1.73 Å by molecular replacement, presenting a correlation coefficient of 0.558 and an R factor of 0.439.
The protein C-phycocyanin, involved in photosynthesis, has been purified from three cyanobacterial species: Spirulina, Phormidium and Lyngbya. These three proteins have been crystallized and characterized using X-ray crystallography.
The expression and crystallization of recombinant E. coli YeaZ protein, which belongs to a family of proteins of unknown function that are conserved in Gram-positive and Gram-negative bacteria, is reported. Phasing of the diffraction data was performed by the MAD method using the anomalous signal of a gadolinium complex, Gd-DOTMA.
The (cytosine-5)-DNA methyltransferase NlaX from N. lactamica has been crystallized in space group P321, with unit-cell parameters a = 121.98, b = 121.98, c = 56.71 Å.
Single crystals of the central structure domains from mumps virus F protein have been obtained by the hanging-drop vapour-diffusion method. A diffraction data set has been collected to 2.2 Å resolution.
Mouse L1 modules Ig I–IV and F3 I–III were crystallized. The crystals diffracted X-rays to 3.5 and 2.8 Å resolution, respectively.
The ammonium transporter Amt-1 from the cytoplasmic membrane of the hyperthermophilic archaeon A. fulgidus has been purified and crystallized.
Purification, crystallization and preliminary X-ray analysis of a hexameric β-glucosidase from wheat
Recombinant β-glucosidase from wheat seedlings complexed with a substrate aglycone has been crystallized in a hexameric active form. A diffraction data set has been collected at 1.7 Å.


 journal menu
journal menu


























![[publBio]](http://journals.iucr.org/logos/publbio.gif)





