issue contents
December 2010 issue
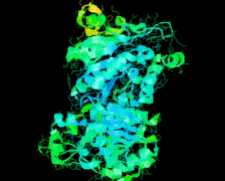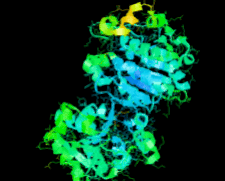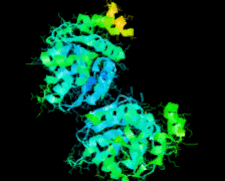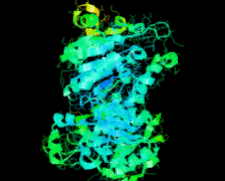
Cover illustration: Structure of Hsp33/YOR391Cp from the yeast Saccharomyces cerevisiae, p. 1557.
editorial
Free 

The problem of undercounting of citations that are published only in supplementary material is studied for the journals Nature, Science, Cell and the Proceedings of the National Academy of Sciences (USA).
structural communications
A 1.35 Å resolution crystal structure of Cas2 from the bacterium Desulfovibrio vulgaris (DvuCas2) is reported.
PDB reference: Cas2, 3oq2
The crystal structure of Hsp33 from S. cerevisiae was solved at 2.40 Å resolution. Structural comparison showed that Hsp33 has a very similar structure to Hsp31, except for some deviations in helices α2–α3 of the core domain.
PDB reference: Hsp33/YOR391Cp, 3mii
Crystal structures of biosynthetic arginine decarboxylase (ADC, SpeA) from E. coli and C. jejuni are reported.
The crystal structure of human PI3K p85β iSH2 domain has been determined to 3.3 Å resolution. Comparison with the published structure of the bovine p85β iSH2 domain bound to the influenza A virus nonstructural protein 1 indicates that little or no structural change occurs upon complex formation. Structural analysis of human and bovine p85β iSH2 domains reveals conformational plasticity in the interhelical turn region of the coiled-coil motif.
The structure of human vascular adhesion protein 1 in space group C2 with seven molecules in the asymmetric unit has been refined at a resolution of 2.9 Å.
PDB reference: human vascular adhesion protein 1, 3ala
crystallization communications
The novel esterase Rv0045c from M. tuberculosis was expressed and purified to homogeneity. The crystals of native and SeMet-labelled Rv0045c protein that were obtained diffracted to resolutions of 2.7 and 3.0 Å, respectively.
A recombinant multiple cofactor-dependent DNA ligase from S. zilligii has been purified and crystallized. X-ray diffraction data were collected to 2.9 Å resolution and the crystals belonged to space group P1.
The cloning, purification and crystallization of the E. coli lipoproteins BamC, BamD and BamE is reported. X-ray diffraction data at high resolution were obtained for each of the proteins or protein domains.
The purification, crystallization and preliminary crystallographic analysis of the C-terminal peptide-binding domain of the human Hsp40 Hdj1 and of full-length Hdj1 are reported.
The F. tularensis protein IglE from the FPI, which is a component of the type VI-like secretion system, has been crystallized and preliminary X-ray data have been collected.
N. tabacum class V chitinase has been crystallized. X-ray diffraction data were collected to 1.2 Å resolution using a synchrotron-radiation source.
Recombinant calcineurin B-like protein 1 from Ammopiptanthus mongolicus (AmCBL1) was overexpressed, purified and crystallized.
Recombinant thiol peroxidase from Y. pseudotuberculosis has been purified and crystallized in three crystal forms.
The receptor-binding domain of botulinum neurotoxin serotype D was expressed in E. coli using a codon-optimized cDNA. The highly purified protein crystallized in space group P212121, with unit-cell parameters a = 60.8, b = 89.7, c = 93.9 Å, and the crystals diffracted to 1.65 Å resolution.
In this study, a recombinant C-terminally truncated form of D. radiodurans DdrA (DdrA157) comprising the first 157 residues of DdrA has been expressed in Escherichia coli, purified and crystallized.
Streak-seeding markedly improved the stability, crystal-growth rate and diffraction quality of dilated-cardiomyopathy-associated mutant metavinculin crystals.
Open  access
access
 access
accessWzi is a membrane protein from E. coli thought to be involved in the attachment of capsular polysaccharides to the bacterial surface. This reports describes recombinant Wzi's purification, crystallization and the results of initial diffraction studies.
Information about the intermediate states in the oligomerization process of a pore-forming toxin is lacking. Two oligomerization-deficient mutants of aerolysin, a major virulence factor secreted by A. hydrophila, which is involved in gastrointestinal infections in humans, have been crystallized in their proteolyzed forms and their preliminary X-ray analysis is reported.
The coupling region NBD94674–781 of Py235 from P. yoelii was cloned, overexpressed, purified and crystallized. The crystals belonged to space group C2 and diffracted to 2.9 Å resolution using synchrotron radiation.
AgaB from Pseudoalterromonas sp. CY24 has been crystallized. The crystals of wild-type enzyme and the selenomethionine derivative diffracted to 1.97 and 2.64 Å, respectively.
The preparation and successful crystallization of the Grb7 SH2 domain in complex with the specific cyclic peptide inhibitor G7-18NATE are reported. This structure is anticipated to reveal the basis of the binding affinity and specificity and to assist with the development of second-generation inhibitors of Grb7, which is involved in cancer progression.
The crystallization of a specific nanobody against mouse PrPC and preliminary diffraction analysis of a crystal that diffracted to 1.23 Å resolution are presented.
Zebrafish Drgal1-L2 was expressed in E. coli, purified and crystallized in the presence of lactose. X-ray diffraction data have been collected to a resolution of 1.5 Å.
Diffraction from crystals of the cobalamin methyltransferases CobJ, CobM, CobF and CobL (full-length and C-terminal domain) from R. capsulatus is reported; three of these enzymes also catalyse the auxiliary reactions of ring contraction (CobJ), decarboxylation (CobL) and deacylation (CobF).
The ferritin homolog, BfrB (Rv3841), from Mycobacterium tuberculosis has been purified, crystallized, and diffraction data were collected to 2.5 Å resolution. Here, preliminary crystallographic characterization and SAXS analyses are reported.
A secreted lectin from M. tuberculosis has been crystallized.
The C-terminal fragment of Streptococcus agalactiae (group B streptococcus) major (backbone) pilin GBS80 was purified and crystallized in two different space groups. Single-wavelength anomalous dispersion (SAD) data collected to 2.0 Å resolution on a iodide (NaI) derivative crystal using the home source were used to obtain initial phases.
Nattokinase, a protein found in high levels in the traditional Japanese food natto, has been reported to have high thrombolytic activity. In the present study, the crystallization of native nattokinase and the collection of X-ray diffraction date from a nattokinase crystal to a resolution of 1.74 Å are reported.
The N-terminal domain of the DEAD-box helicase from S. aureus strain Mu50 was cloned, overexpressed, purified and crystallized. X-ray diffraction data were collected to 2.60 Å resolution.
The crystallization of the six-subunit Na+-NQR complex from V. cholerae is reported.
NADPH-dependent 5-keto-D-gluconate reductase from G. suboxydans IFO12528 (5KGR) was expressed, purified and crystallized with 5-keto-D-gluconate and NADPH using the sitting-drop vapour-diffusion method. Crystals of the 5KGR–NADPH complex and of the 5KGR–NADPH–5-keto-D-gluconate complex diffracted X-rays to 1.75 and 2.26 Å resolution, respectively.
addenda and errata
Free 

Free 



 journal menu
journal menu













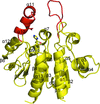



























![[publBio]](/logos/publbio.gif)






