issue contents
May 2011 issue
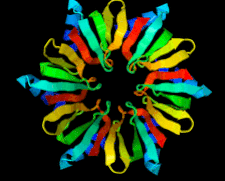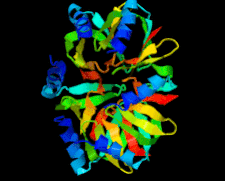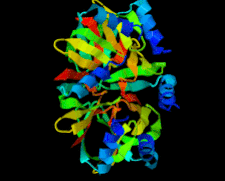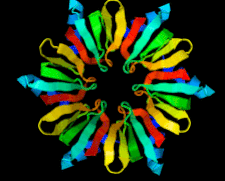
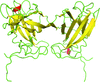
Cover illustration: Full-length host factor Q![[beta]](/logos/entities/beta_rmgif.gif) (Hfq) from Escherichia coli (Beich-Frandsen et al., p. 536).
(Hfq) from Escherichia coli (Beich-Frandsen et al., p. 536).
structural communications
The crystal structure of C. thermocellum CelK CMB4 confirms unique extended binding pocket features of clostridial CBM4s.
PDB reference: cellulase K CBM4, 3p6b
Here, the crystal structure of mature fHbp determined at 2 Å resolution is presented and is compared with the structure of the same protein in complex with factor H domains 6 and 7 recently solved using X-ray techniques.
PDB reference: fHbp, 3kvd
The full-length RNA-chaperone Hfq from Escherichia coli (102 amino acids) has been crystallized. The crystal belongs to the P1 space group. The structure was solved to 2.85 Å resolution, revealing that the disordered C-terminal resides on the proximal face of the hexamer.
PDB reference: Hfq, 3qhs
The X-ray structure of phosphopantetheine adenylyltransferase (PPAT) from M. tuberculosis in complex with its feedback regulator coenzyme A (CoA) was determined to 2.11 Å resolution. Unlike previous X-ray structures of PPAT–CoA complexes from other bacteria, which showed two distinct conformations of bound CoA, only one conformation of bound CoA is observed in the M. tuberculosis PPAT–CoA complex.
PDB reference: PPAT–CoA complex, 3pnb
crystallization communications
Here, the recombinant ARE-binding region of HuR (residues 18–186) was crystallized in space group P21212, with unit-cell parameters a = 41.2, b = 133.1, c = 31.4 Å.
The ligand–receptor complex of human growth and differentiation factor 5 (GDF5) bound to the extracellular domain of human BMP type IA receptor (BRIAEC) can be crystallized as long as the GDF5 variant R57A, which exhibits an enhanced affinity for BRIA, is employed.
An engineered deletion of a nonconserved loop in the RUN domain of Rab6-interacting protein 1 enhances the solubility of the recombinant protein when expressed in E. coli. Crystals of the engineered effector in complex with Rab6a diffracted X-rays to 3.25 Å resolution.
The sequence design, purification, crystallization and preliminary X-ray diffraction analysis of the novel cap-independent translation RNA element from the 3′ untranslated region of the barley yellow dwarf virus genome is reported.
Solving the structure of MjNifH1 may help in better understanding its function and may supply some clues to understanding the evolution of nitrogenase. The full-length protein with an additional His6 tag at the C-terminus was expressed, purified and crystallized by the hanging-drop vapour-diffusion method at 287 K.
A crystal of a swine MHC class I molecule diffracted to 2.2 Å resolution and belonged to space group P212121, with unit-cell parameters a = 66.1, b = 74.1, c = 98.6 Å; it contained one molecule in the asymmetric unit. The Matthews coefficient and the solvent content were calculated to be 2.74 Å3 Da−1 and 55.17%, respectively.
Sm23, a novel SGNH arylesterase from S. meliloti 1021, was crystallized in space group I4122 and diffraction data were collected to a resolution of 2.2 Å.
Mushroom tyrosinase was crystallized in two different space groups. The crystals diffracted to 3.0 Å resolution (P21) or 2.6 Å resolution (P21212).
Phycocyanin and phycoerythrin from P. yezoensis Ueda were prepared using co-precipitation with ammonium sulfate and hydroxyapatite column chromatography with improved purity and yield. The preliminary X-ray analysis clarified the ambiguity in the unit-cell contents resulting from the Matthews coefficient calculation and the final model showed that phycocyanin is present as two α6β6 heterododecamers in the asymmetric unit associated by an NCS twofold axis.
The transaldolase enzyme from T. acidophilum has been crystallized in two different space groups.
In order to begin an exact determination of hydrogen positions in proteins, a neutron diffraction study of bovine gamma-chymotrypsin has been conducted. This paper details the data collection of the protein at pD (pH*) 7.1.
Diffraction-quality crystals of N. equitans neelaredoxin have been produced. The expression, purification and crystallization of the protein and preliminary X-ray crystallographic analysis of the crystals are reported.
The mannose-binding lectin domain of MSMEG_3662 from M. smegmatis has been crystallized and preliminary X-ray studies have been carried out on the crystals.
A CAL PDZ-domain construct was crystallized in complex with the selective peptide inhibitor iCAL36, which has been shown to enhance ΔF508-CFTR chloride-channel activity in human airway epithelial cells.
The V. parahaemolyticus resuscitation-promoting factor YeaZ has been purified and crystallized by the hanging-drop vapour-diffusion method. A diffraction data set has been collected to 3.1 Å resolution.
The purification, preliminary characterization and crystallization of a lectin purified from M. indica latex are presented. Using synchrotron radiation, a diffraction data set to 2.8 Å resolution was collected.
The N-terminal domain of thioredoxin-interacting protein has been expressed, purified and crystallized. The crystals belonged to a monoclinic space group and diffracted to 3 Å resolution using synchrotron radiation.
Recombinant purine nucleoside phosphorylase from B. subtilis strain 168 has been crystallized at different pH values using the sitting-drop vapour-diffusion method.
Cloning, expression, purification and X-ray data collection of the RadA intein from the hyperthermophilic archaebacterium P. horikoshii are reported.
The gene product Rv3168, a putative aminoglycoside phosphotransferase (APH), from Mycobacterium tuberculosis has been crystallized using the sitting-drop vapour-diffusion method and X-ray diffraction data were collected to a maximum resolution of 1.67 Å on a synchrotron beamline.
Here, the overexpression, purification and crystallization of the isolated C39 peptidase-like domain of the Escherichia coli ABC transporter haemolysin B are described.
The crystallization of the precursor of a thermostable variant of papain and the collection of diffraction data to 2.6 Å resolution are reported.
The crystallization of recombinant lidA, a translocated substrate of the Legionella pneumophila Dot/Icm type IV secretion system, is reported. Crystals were obtained and diffracted to 2.75 Å in space group P212121.


 journal menu
journal menu

































![[publBio]](/logos/publbio.gif)





