issue contents
July 2011 issue
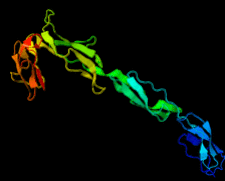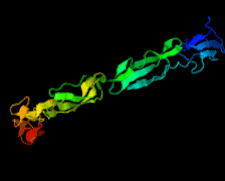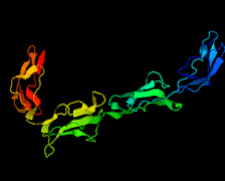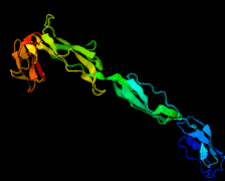
Cover illustration: Structure of the rat complement regulator CrrY (Roversi et al., p. 739).
structural communications
The crystal structure of POTRA4–5 has been determined to 1.50 Å resolution with an R factor of 14.7% and an Rfree of 18.9%.
PDB reference: BamA POTRA4–5, 3q6b
Open  access
access
 access
accessThe structure of rat CrrY1–4 determined in two distinct crystal forms shows a pronounced bend at the interface between domains 3 and 4.
The crystal structure of Drosphila melanogaster Rab6 bound to the non-hydrolysable GTP analogue GMPPNP has been determined at a resolution of 1.4 Å.
PDB reference: Rab6 GTPase, 2y8e
The crystal structure of the bacterial protein LpxD from P. aeruginosa was solved and refined at 1.3 Å resolution. The overall domain architecture and biological assembly are similar to those found in previously solved structures of LpxD from other species.
PDB reference: LpxD, 3pmo
The crystal structure of an extremely thermostable multicopper oxidase from a hyperthermophile was determined.
PDB reference: multicopper oxidase, 3aw5
crystallization communications
The DIX domain of Ccd1 was purified and crystallized.
Open  access
access
 access
accessCwp19 is a putatively surface-located protein from Clostridium difficile. A recombinant N-terminal protein (residues 27–401) lacking the signal peptide and the C-terminal cell-wall-binding repeats (PFam04122) was crystallized using the sitting-drop vapour-diffusion method and diffracted to 2 Å resolution.
The design, expression and purification of receptor protein tyrosine phosphatase γ and two mutants and their crystallization in three different crystal forms are described.
A novel ferredoxin/thioredoxin reductase-like protein from M. acetivorans was heterologously expressed in E. coli, purified and then subjected to crystallization. Preliminary X-ray diffraction studies revealed that the crystal belonged to a primitive cubic space group, with unit-cell parameters a = b = c = 92.72 Å.
Highly thermostable β-1,3-xylanase from T. neapolitana strain DSM 4359 was prepared and crystallized. X-ray diffraction data were collected to a resolution of 1.82 Å.
A hyperthermophilic nucleotidyltransferase encoded by the TTHA1015 gene from T. thermophilus was crystallized. X-ray diffraction data were collected to 1.7 Å resolution.
Open  access
access
 access
accessThe α-mannosidase I inhibitor kifunensine inhibited N-glycan processing in long-term cultures of Chinese hamster ovary cells, allowing deglycosylation and crystallization of the homodimeric extracellular region of the inhibitory glycoprotein receptor CTLA-4 (CD152).
The C-terminal domain of SARS coronavirus nonstructural protein 2 was cloned, overexpressed, purified and crystallized; the crystals diffracted to 2.5 Å resolution.
Recombinant GK1506 from the thermophilic bacterium Geobacillus kaustophilus has been expressed, purified and crystallized. A 2.6 Å resolution native data set was collected from a single flash-cooled crystal.
The substrate binding protein (SP_0149) of an ABC transporter from Streptococcus pneumoniae was molecularly cloned, overexpressed and purified. Diffraction quality crystals were grown using the hanging-drop vapour-diffusion technique.
The ice-binding protein from Leucosporidium sp. AY30 was cloned, expressed, purified and crystallized. A complete data set was collected to 1.5 Å resolution.
The putative sensor histidine kinase domain of the cytoplasmic protein HksP4 from the hyperthermophilic bacterium A. aeolicus VF5 was expressed, purified and crystallized by the sitting-drop vapour-diffusion method. Crystals were obtained in the presence of ATP and AMPPNP; they were found to belong to the same space group P212121 and diffracted X-rays to 3.1 and 2.9 Å resolution, respectively.
The cytoplasmic domain of FlhB from S. typhimurium has been purified and crystallized.
MsDpo4 from M. smegmatis, a member of the Y-family of DNA polymerases, was expressed and purified to homogeneity. Crystals of native MsDpo4 in the apo state were obtained and X-ray diffraction data were collected to a maximum resolution of 2.6 Å.
The crystallization of a putative SCP-x thiolase from M. smegmatis is described.
The 4-diphosphocytidyl-2-C-methyl-D-erythritol kinase (IspE) from M. tuberculosis H37Rv was overexpressed in E. coli, purified and crystallized. Diffraction data for the native enzyme were collected to 2.1 Å resolution.
Shikimate dehydrogenase from Thermotoga maritima has been overexpressed, crystallized, and its crystal structure has been determined. X-ray diffraction data have been collected to 1.45 Å.
Membrane-bound respiratory [NiFe] hydrogenase was purified from H. marinus and crystallized. A diffraction data set was collected to 1.25 Å resolution.
Crystallization of extracellular dermal glycoprotein and the inhibition complex that it forms with endo-β-glucanase.
The large pentamodular C. thermocellum cellulosomal arabinoxylanase CtXyl5A (without the C-terminal dockerin) has been crystallized and data have been collected to a resolution of 2.64 Å.


 journal menu
journal menu











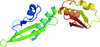





















![[publBio]](/logos/publbio.gif)





