issue contents
July 2012 issue
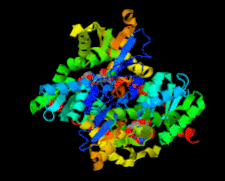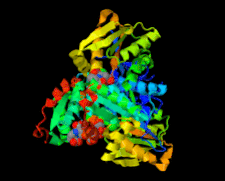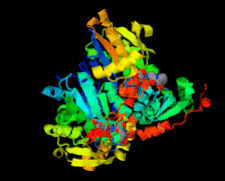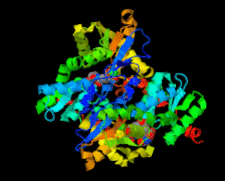
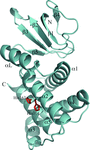
Cover illustration: Structure of Leishmania major cysteine synthase (Fyfe et al., p. 738).
structural communications
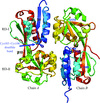
The crystal structure of the regulatory domain of NMB2055, a putative MetR regulator, was solved at 2.5 Å resolution.
Open  access
access
 access
accessA crystallographic and biochemical study of L. major cysteine synthase, which is a pyridoxyl phosphate-dependent enzyme, is reported. The structure was determined to 1.8 Å resolution and revealed that the cofactor has been lost and that a fragment of γ-poly-D-glutamic acid, a crystallization ingredient, was bound in the active site. The enzyme was inhibited by peptides.
PDB reference: cysteine synthase, 4air
The 1.85 Å resolution structure of the signal transduction protein TRAP is presented. The overall fold of TRAP is an unsymmetrical eight-stranded β-barrel with five helices.
PDB reference: TRAP, 4ae5
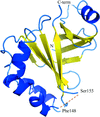
The crystal structure of the RBD-PRDI fragment of the antiterminator protein GlcT from Bacillus subtilis has been solved at 2 Å resolution. The structure represents an inactive state of the protein.
PDB reference: RBD-PRDI fragment of GlcT, 3rio
crystallization communications
In this work, Pz peptidase B, an intracellular M3 metallopeptidase that is found in the thermophile Geobacillus collagenovorans MO-1, was crystallized using the counter-diffusion method.
The complex of CCM3 and the C-terminal domain of MST4 has been successfully constructed, purified and crystallized. The crystal diffracted to a resolution of 2.4 Å.
The JmjC domain-containing histone demethylase NO66 from H. sapiens was overproduced in E. coli, purified and crystallized. Diffraction data were collected to 2.29 Å resolution.
A novel feruloyl esterase (EstF27) identified from a soil metagenomic library has been crystallized and a complete data set was collected from a single cooled crystal using an in-house X-ray source.
An atypical short-chain dehydrogenase/reductase from Vibrio vulnificus was expressed, purified and cocrystallized with NADPH by the sitting-drop vapour-diffusion method. X-ray diffraction data were collected to 1.80 Å.
This paper reports the cloning, expression, purification, crystallization and preliminary X-ray crystallographic study of acyl-protein thioesterase 1 from S. cerevisiae.
The extracellular region of mouse Enpp1 was expressed, purified and crystallized. An X-ray diffraction data set was collected to 3.0 Å resolution by employing a helical data-collection strategy involving a micro-focus synchrotron beam.
The crystallization and preliminary X-ray diffraction analysis at 1.25 Å resolution of free-ligand arginine kinase from the Pacific whiteleg shrimp Litopenaeus vannamei are reported. Crystals belong to space group P212121, phases were determined by molecular replacement and refinement was performed with Phenix.
The cloning, overexpression, purification, crystallization in a triclinic space group and preliminary X-ray diffraction analysis of the high-molecular-weight ketoacyl reductase FabG4 complexed with NADH are reported.
Approximately five decades have passed with only one or two new antibiotics making it into clinical use. Phosphoglycerate kinase from A. baumanii has been selected as a potential target for antibiotic development; this paper presents the initial structural biological results from this research.
The putative adhesion domain of the multidomain protein Epf from S. pyogenes has been crystallized in space groups P21 and P212121. The crystals diffracted to 2.0 and 1.6 Å resolution, respectively, at the Australian Synchrotron.
The GhKCH2 motor domain was crystallized and the pH of the crystallization buffer was shown to have a significant effect on the crystal morphology and diffraction quality.
The overexpression, purification, crystallization and preliminary X-ray diffraction analysis of protein elicitor PevD1 from Verticillium dahliae are reported.
The ice-binding protein FfIBP from F. frigoris PS1 was overexpressed, purified, characterized and crystallized, and preliminary X-ray crystallographic analysis was performed.
The S. epidermidis carrier protein DltC has been crystallized in order to elucidate the functional role of DltC in the alanylation of lipoteichoic acids in bacteria.
The crystallization of the human NORE1 SARAH domain is reported.
Open  access
access
 access
accessThe expression, purification and crystallization of an N-terminal fragment of SHARPIN are reported. Diffraction-quality crystals were obtained using a two-dimensional grid-screen seeding technique.
The yeast Epsin-1 (ent1) gene has been cloned and expressed in Escherichia coli. The protein product of a construct containing the ENTH-UIM modules was purified to homogeneity and subjected to crystallization screening using the sitting-drop vapour-diffusion method. Refined conditions containing polyethylene glycol 3350 and Tacsimate yielded thin rod-like crystals.
A hydroxycinnamoyl-CoA shikimate/quinate hydroxycinnamoyltransferase involved in chlorogenic acid biosynthesis in C. canephora was crystallized using the vapour-diffusion method. A diffraction data set was collected to 3.0 Å resolution on the microfocus beamline (ID23-2) at ESRF and a structure solution was obtained using molecular replacement.
The purification, crystallization and preliminary X-ray diffraction analysis of the C-terminal domain of δ-COP, a medium-sized subunit of the COPI complex, are reported.
To characterize the ISP family of proteins present in apicomplexan parasites, ISP1 from T. gondii was expressed, purified and crystallized. Two crystal forms (cubic and orthorhombic) were analyzed by X-ray diffraction and data were processed to 2.05 and 2.1 Å resolution, respectively.
Open  access
access
 access
accessNa-FAR-1, a fatty acid- and retinol-binding protein, was expressed in bacteria, purified and crystallized. Crystals grew in two different morphologies under the same conditions.
A protease involved in turnover of a photosynthesis reaction centre protein has been crystallized and preliminary diffraction analysis was performed.
Construct engineering and crystallization of E. coli PgaB using in situ proteolysis and mass spectrometry is reported.


 journal menu
journal menu











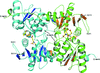





















![[publBio]](/logos/publbio.gif)




