issue contents
May 2013 issue
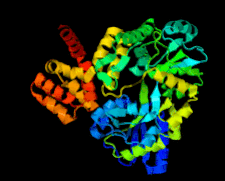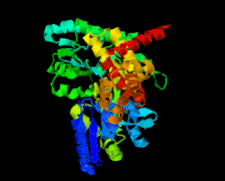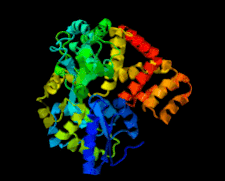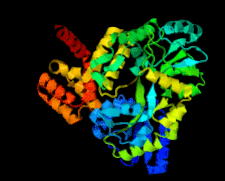
Cover illustration: Structure of the CARD8 caspase-recruitment domain (Jin et al., p. 482).
structural communications
The high-resolution crystal structure of the complex between the catalytic domain of karilysin and the tetrapeptide SWFP has been determined.
PDB reference: Kly18–SWFP complex, 4in9
Here, the crystal structure of Yersinia pestis tip protein LcrV is reported at a resolution of 1.65 Å.
PDB reference: Yersinia pestis tip protein LcrV, 4jbu
The structure of the CARD8 caspase-recruitment domain suggests that its regulatory functions may be mediated by electrostatic attractions between the CARD8 CARD and its partner domains.
PDB reference: CARD8 CARD, 4ikm
Analysis of the different redox states of the catalytic cysteines in four crystallographic structures of thioredoxin 1 from the Pacific whiteleg shrimp L. vannamei highlights their reactivity and corroborates the existence of a structural dimer mediated by an interface of 12 residues which includes a disulfide bridge between the Cys73 residues of each monomer.
PDB references: reduced LvTrx, 4aj6; partially reduced LvTrx, 4aj8; thioredoxin, 3zzx; oxidized LvTrx, 4aj7
The cocrystal structure of the PTB domain of integrin cytoplasmic domain-associated protein-1 (ICAP1) in complex with a 29-residue fragment of Krev/Rap1 interaction trapped-1 (KRIT1) has been determined.
PDB reference: ICAP1–KRIT1170–198, 4jif
Crystals of E. coli triosephosphate isomerase were obtained as a contaminant and its structure was determined to 1.85 Å resolution.
PDB reference: triosephosphate isomerase, 4iot
crystallization communications
P. aeruginosa UDP-MurNAc-tripeptide:D-Ala-D-Ala ligase (MurF) was crystallized in the presence of substrate and diffraction data were collected to 1.92 Å resolution.
The expression, purification, crystallization and preliminary crystallographic characterization of wild-type and L(M196)H-mutant reaction centres of Rba. sphaeroides strain RV are reported.
The crystallization and preliminary X-ray diffraction analysis of the strawberry PR-10 proteins Fra a 1E and Fra a 3 in the presence of the natural flavonoid (+)-catechin are presented.
The 3-hydroxyacyl-CoA dehydrogenase from Ceanorhabditis elegans was overexpressed, purified and crystallized in two different space groups (P1 and P212121). The molecular dimers found in one asymmetric unit from both space groups are identical but each packs in a distinct manner.
A streptococcal domain of unknown function 1792 has been crystallized.
Tse3, one of the effectors of the type VI secretion system in Pseudomonas aeruginosa, has been crystallized and diffracted to 1.5 Å resolution.
Aldehyde dehydrogenase (ALDH) catalyses the oxidation of aldehydes using NAD(P)+ as a cofactor. The aldh gene from B. cereus was cloned; the protein was expressed, purified and crystallized, and a preliminary X-ray crystallography analysis was performed.
Triclinic crystals of YhgD from B. halodurans have been obtained. X-ray data have been collected to 1.9 Å resolution using synchrotron radiation.
The L. donovani coronin CRN12 coiled-coil domain (5.8 kDa) was cloned, overexpressed and purified to homogeneity. Crystals of recombinant L. donovani coronin CRN12 coiled-coil domain were grown by vapour diffusion using a hanging-drop setup.
The complex of the abscisic acid receptor PYL3 with (−)-ABA was crystallized and refined to obtain high-quality diffraction data. Diffraction data were collected and processed at 2.65 Å resolution.
The lumenal domain of Erv41p from S. cerevisiae has successfully been expressed, purified and crystallized. X-ray diffraction data were collected to a resolution of 2.0 Å.
A core fragment of Salmonella FlgG has been expressed, purified and crystallized, and X-ray diffraction data sets were collected to 2.0 Å resolution.
An N-terminally truncated mutant of the colicin E7 nuclease domain was crystallized and diffraction data set was collected to 1.6 Å resolution.
Crystallization and in-house SAD phasing unveils the structure of a unique thermophilic Rieske ferredoxin with a stabilizing disulfide bridge.
The C-terminal LRR domain of S. pyogenes protein 0843 was overexpressed in E. coli, purified and crystallized. A complete data set to 1.59 Å resolution was collected using synchrotron radiation.
The Escherichia coli cyclic AMP receptor protein (CRP) crystals were obtained and diffracted at a 2.9 Å resolution, which belonged to the space group P3121, with unit-cell parameters a = b = 76.03, c = 144.00 Å. The asymmetric unit was found to contain one protein molecule and a half 38 bp full-length double-stranded DNA fragment with a Matthews coefficient of 2.62 Å3 Da−1 and a solvent content of 53.14%.
The cloning, expression, purification, crystallization and preliminary X-ray diffraction analysis of MSMEG_5817 is described.
EaLsc, a levansucrase from E. amylovora, has been cloned, expressed, purified and crystallized. X-ray crystallographic analysis and data collection to 2.77 Å resolution have been carried out. The structure was solved by molecular replacement and refinement is in progress.
The expression, purification and crystallization of Omp6, a protoilludene synthase from O. olearius, are described.
The pyridoxal biosynthesis lyase (PdxS) from Mycobacterium tuberculosis was crystallized and diffracted to a resolution of 1.8 Å. The crystals were found to belong to space group I222 or I212121, with unit-cell parameters a = 110.75, b = 126.08, c = 180.82 Å, α = β = γ = 90°.
The cloning, purification, crystallization and preliminary crystallographic analysis of the effector-binding domain of the transcriptional regulator AlsR from B. subtilis are described.
The Cmr2–Cmr3 subcomplex from P. furiosus was co-crystallized with 3′-AMP. X-ray diffraction data for the crystals were collected to 2.6 Å resolution using a synchrotron-radiation source.
The xylose isomerase enzyme from B. adolescentis was expressed in E. coli, purified and crystallized. A native X-ray diffraction data set was collected to 1.7 Å resolution.


 journal menu
journal menu












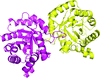



















![[publBio]](/logos/publbio.gif)





