issue contents
June 2013 issue
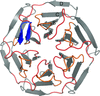
Cover illustration: Structure of PatF from Prochloron didemni (Bent et al., p. 618).
structural communications
A mutant of the Kelch domain of the human Keap1 protein has been designed in order to enable the soaking of small-molecule ligands. The apo structure of this mutant is reported at 1.98 Å resolution and the suitability of the crystal system has been demonstrated by the structure of the mutated Keap1 Kelch domain in complex with a cyclic peptide derived from Nrf2.
The structure of DszC from R. erythropolis DS-3 was solved using the single-wavelength anomalous dispersion method.
PDB reference: DszC, 4jek
The triclinic structure of the HIV-1 capsid protein contains four molecules in the asymmetric unit that form a novel packing interface that could conceivably resemble an intermediate structure that is involved in the early steps of HIV-1 assembly.
PDB reference: carboxy-terminal domain of HIV-1 capsid protein, 4ipy
The crystal structure of the type VI secretion-system protein TssJ1 from P. aeruginosa was solved by iodide SAD at a resolution of 1.4 Å.
PDB reference: TssJ1, 3zhn
Open  access
access
 access
accessThe structure of a bifunctional deaminase/reductase involved in riboflavin biosynthesis in the pathogen A. baumannii has been determined in two crystal forms.
Open  access
access
 access
accessThe X-ray crystal structure of PatF from P. didemni was solved by the single-wavelength anomalous diffraction method to a resolution of 2.13 Å.
PDB reference: PatF, 4bg2
Open  access
access
 access
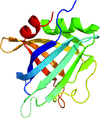
accessThe structure of ErpC, a member of the complement regulator-acquiring surface protein family from B. burgdorferi, has been solved, providing insights into the strategies of complement evasion by this zoonotic bacterium and suggesting a common architecture for other members of this protein family.
PDB reference: ErpC, 4bf3
Open  access
access
 access
accessB. burgdorferi binds complement factor H using a dimeric surface protein, CspA (BbCRASP-1). Presented here is a new structure of CspA that suggests that there is a degree of flexibility between subunits which may have implications for complement regulator binding.
PDB reference: CspA, 4bl4
The crystal structure of an A-form RNA duplex obtained by degradation of 6S RNA in a crystallization droplet has been solved. The role of a ribose-zipper motif in the intermolecular packing is presented.
PDB reference: A-form DNA, 4jrt
crystallization communications
The crystal of E. coli 23S rRNA methyltransferase RlmM in this study belongs to a new space group (P21) and is different from the known P31 or P3121 space groups.
A primitive monoclinic crystal of the rhesus macaque MHC class I molecule Mamu-B*17 complexed with an SIVmac239 Env peptide was obtained and belonged to space group P2, with unit-cell parameters a = 68.3, b = 45.0, c = 81.5 Å, β = 96.5°. The crystal diffracted to 2.55 Å resolution.
An osmotin from the latex of C. procera has been crystallized in both tetragonal and trigonal forms suitable for structure determination.
AAL-2, an A. aegerita lectin that specifically recognizes nonreducing terminal acetylglucosamine, was crystallized and preliminary crystallographic studies were performed on this protein. This work provides a solid foundation for determination of the AAL-2 structure and illustration of the sugar-binding specificity of this novel lectin.
Dihydrodipicolinate reductase has been cloned, expressed and crystallized. The crystals belonged to the orthorhombic space group P222, with unit-cell parameters a = 80.0, b = 100.8, c = 147.6 Å.
GluB, a substrate-binding protein from C. glutamicum, was expressed, purified and crystallized, followed by X-ray diffraction data collection and preliminary crystallographic analysis.
A thermostable alanine racemase from T. tengcongensis MB4 was expressed in E. coli and purified. Crystallization and preliminary X-ray crystallographic analysis were performed for the recombinant enzyme.
The flagellar motor `brake' protein YcgR from E. coli was crystallized with c-di-GMP. The crystals diffracted to 2.3 Å resolution and belonged to space group R3:H, with unit-cell parameters a = b = 93.96, c = 109.61 Å.
Crystals of EtFPOX from E. terrenum sp. diffracted to 1.9 Å resolution and belonged to space group P212121, with unit-cell parameters a = 65.6, b = 80.0, c = 83.4 Å.
An additional example in which heterogeneous nucleation has helped in the search for crystallization conditions of a protein is reported. Optimization of the crystallization conditions led to the formation of single crystals of γ-glutamyl transpeptidase from B. licheniformis that diffracted to about 3.0 Å resolution.
The cloning, expression, purification and crystallization of hyaluronate lyase from S. suis are reported.
X. campestris pv. campestris xyloglucanase was produced in E. coli using recombinant pSMT3 vector. Crystals were obtained and X-ray data sets were collected for both the apoenzyme and its complex with glucose.
The ATPase domain of M. tuberculosis DNA gyrase was crystallized using hanging-drop vapour diffusion. The crystals belonged to space groups P1 and P21. Diffraction data were collected to resolutions of 2.9 and 3.3 Å, respectively.
The novel haloalkane dehalogenases DpcA from P. cryohalolentis K5 and DmxA from Marinobacter sp. ELB17 were successfully crystallized and diffraction data were collected to resolutions of 1.05 and 2.49 Å, respectively.
The DNA-binding domain of the response regulator SaeR was crystallized. Determination of its structure will reveal the residues that are critical for protein–DNA recognition.
The recombinant tetrathionate hydrolase from the acidophilic chemolithoautotrophic bacterium A. ferrooxidans was refolded for activation, purified and crystallized. Diffraction data were collected to 2.15 Å resolution.
A GH27 arabinopyranosidase from G. stearothermophilus (Abp) has been crystallized in the primitive orthorhombic space group P212121. Full diffraction data sets have been measured for the wild-type enzyme and its D197A catalytic mutant to maximal resolutions of 2.28 and 2.30 Å, respectively, for use in a detailed three-dimensional structural analysis of the Abp protein.
The nucleocapsid protein from Tomato spotted wilt virus has been crystallized as a trimer. The crystal diffracted to resolution of 2.7 Å.
laboratory communications
Crystals of recombinantly expressed ρ-Da1a toxin from eastern green mamba that were suitable for structure determination were discovered on defrosting a partially lyophilized sample.
PDB reference: Gly-ρ-Da1a-K34A, 4iye
addenda and errata
Free 



 journal menu
journal menu












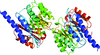



















![[publBio]](/logos/publbio.gif)






