issue contents
November 2013 issue
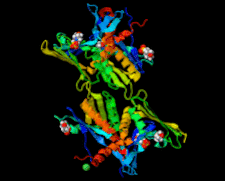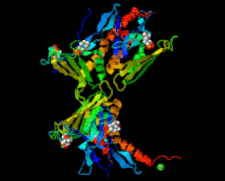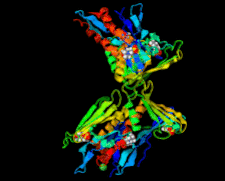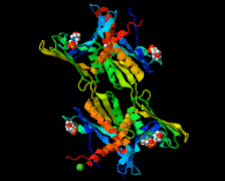
Cover illustration: Whirly protein WHY1 from Arabidopsis thaliana (Cappadocia et al., p. 1207).
structural communications
Open  access
access
 access
accessHere, crystal structures of Ara1 in apo and NADPH-complexed forms are presented at 2.10 and 2.00 Å resolution, respectively.
The crystal structure of CT584 from C. trachomatis is presented at 3.05 Å resolution.
PDB reference: CT584, 4mlk
The tertiary structure of an i-type lysozyme isolated from the common orient clam (Meretrix lusoria)
The tertiary structure of invertebrate-type lysozyme from the common orient clam was determined and compared with that of V. philippinarum lysozyme.
PDB reference: Meretrix lusoria lysozyme, 3ab6
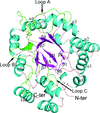
The crystallization and crystal structure determination of the three Whirly proteins from A. thaliana is described.
crystallization communications
The cholesterol-dependent cytolysin listeriolysin O from L. monocytogenes has been purified and crystallized. The orthorhombic crystals diffracted to beyond 2.2 Å resolution and contained one molecule in the asymmetric unit.
The cloning, purification, crystallization and preliminary X-ray diffraction studies of S. aureus homoserine dehydrogenase, an enzyme that regulates the biosynthesis of several essential amino acids, are reported.
The globular cargo-binding domain of human Myo5a was cloned, expressed, purified and crystallized and the crystals were characterized. Special precautions that were necessary to harvest diffraction-quality crystals are described.
The cytoplasmic domain of glutathione peroxidase 3 (GPX3) from A. thaliana was overexpressed in E. coli, purified and crystallized. Diffraction data were collected to a resolution of 2.8 Å.
Crystallization and preliminary X-ray study of alanine dehydrogenase from Bacillus pseudofirmus OF4
An alanine dehydrogenase from B. pseudofirmus OF4 was expressed in E. coli and purified. Crystallization and preliminary X-ray crystallographic analysis of the recombinant enzyme were performed.
ALKBH5 catalyzes the demethylation of N6-methyladenosine in RNA. The ALKBH5 protein was crystallized and X-ray diffraction data were collected to a resolution of 2.4 Å.
A phosphotriesterase-like lactonase dubbed VmoLac isolated from the hyperthermophilic crenarchaeon V. moutnovskia was purified and crystallized. A diffraction data set was collected to 2.4 Å resolution.
Open  access
access
 access
accessCrystal data on a putative portal protein from the thermostable bacteriophage G20C indicate that it forms a 12-subunit assembly.
The VapBC-15 toxin–antitoxin protein complex was purified and crystallized using the vapour-diffusion technique. A diffraction data set was collected to 2.6 Å resolution.
The cloning, expression, purification, crystallization and preliminary X-ray diffraction analysis of the putative transcriptional regulator SPD0280 from S. pneumoniae D39 are described.
The malate dehydrogenase from T. thermophilus has been cloned, overexpressed, purified and crystallized. Crystals belonging to the orthorhombic space group P212121 produced diffraction data to 1.80 Å.
α-Carbonic anhydrase from H. pylori has been purified and crystallized by the hanging-drop vapour-diffusion method. A diffraction data set has been collected to 1.7 Å resolution.
spMCAT from Synechocystis sp. PCC 6803, the first MCAT counterpart from a cyanobacterium, has been successfully cloned, purified and crystallized. A high-quality crystal with good diffraction has been obtained.
The CIDE-N domain of CIDE-3 was purified and crystallized. The crystals were found to belong to space group P32, with unit-cell parameters a = b = 63.35, c = 37.60 Å, γ = 120°. The crystals were obtained at 293 K and diffracted to a resolution of 2.0 Å.
Profilin from the helminthic parasite S. japonicum was expressed, purified and crystallized. The protein was found to be folded and to possess the expected secondary-structure content as determined by circular-dichroism spectroscopy.
The crystallization and preliminary X-ray diffraction analysis of the ribonuclease regulator RraB from E. coli are reported.
MJ0458 protein, an adenylate kinase from Methanocaldococcus jannaschii which is a rich resource of unique enzymes, was crystallized and a set of X-ray diffraction data to 2.70 Å resolution was collected on the beamline BL-17U of the Shanghai Synchrotron Radiation Facility (SSRF). The crystal belonged to space group P41212 or P43212. The unit-cell parameters were a = b = 76.18, c = 238.70 Å, α = β = γ = 90°.
To investigate the molecular basis of immune signalling initiated by the TIR domains of plant disease-resistance proteins, the crystallization and preliminary X-ray diffraction analyses of the TIR domains of three proteins involved in disease resistance in A. thaliana are reported.
The C-terminal coiled-coil domain of SCP3, which is part of the lateral element of the synaptonemal complex, was purified and crystallized. The crystals were found to belong to space group C2, with unit-cell parameters a = 121.29, b = 43.08, c = 57.42 Å, β = 100.71°. The crystals were obtained at 293 K and diffracted to a resolution of 3.2 Å.
The reduced form of the terminal oxygenase component of carbazole 1,9a-dioxygenase from Janthinobacterium sp. J3 was crystallized. The crystals belonged to space group P65 and diffracted to 1.74 Å resolution.
Crystallization and preliminary X-ray studies of a ternary complex consisting of the ribosomal protein L11, the two-domain N-terminal fragment of the ribosomal protein P0 and a specific fragment of 23S rRNA from the archaeon M. jannaschii are reported.
The crystallization and preliminary crystallographic analysis of the putative NlpC/P60 endopeptidase, TTHA0266 is reported. The crystal belonged to space group P61 and the structure was solved with the single-wavelength anomalous dispersion method.
Overexpression,crystallization and preliminary X-ray crystallographic analysis of release factor eRF1-1 from Arabidopsis thaliana.
KatB, a manganese catalase from Anabaena PCC7120 was crystallized and preliminary diffraction data obtained. Crystal belonged to space group P41212 (or enantiomer) and diffracted to 2.2 Å resolution.
The crystallization of acetyl-CoA hydrolase from N. meningitidis is reported.
The catalytic domain of the histidine kinase of the ethylene receptor ETR1 from A. thaliana was expressed and crystallized. The crystals belonged to space group I222 or I212121.
addenda and errata
Free 

Free 

Free 

Free 

A correction is made to the article by Nogly et al. (2012). Acta Cryst. F68, 1113–1115.
Free 

A correction is made to the article by Yu et al. (2013). Acta Cryst. F69, 812–814.


 journal menu
journal menu







































![[publBio]](/logos/publbio.gif)






