issue contents
February 2002 issue
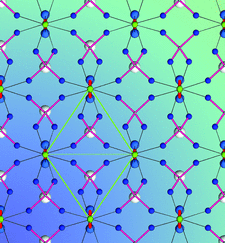
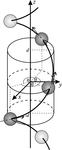
Cover illustration: The crystal structure of titanite, CaTiOSiO4, viewed down the direction of the octahedral TiO6 chains. The full green lines indicate a unit cell of the two-dimensional Ising model, used to predict diffuse scattering intensity observable in layers normal to the chain direction. Ti atoms are shown in green, Ca in light grey, O in blue. The thinner red lines are Si-O bonds, the thick red lines Ti-O1 bonds. Courtesy of T. Malcherek, C. Paulmann, M. C. Domeneghetti & U. Bismayer [J. Appl. Cryst. (2001), 34, 108-113].
research papers
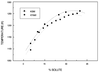
A change in the higher-order structure of the oligomeric protein is directly detectable by a small-angle scattering method. A SAXS study of the denaturation process of the chaperonin protein GroEL showed that the disappearance of the quaternary structure can be monitored by the Kratky plot of the scattered intensities.
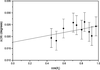
With regard to the application of a commercial focusing device in standard single-crystal X-ray diffractometers, the homogeneity of the beam, the cross section at various distances from the focus and the efficiency of the optic when it is operated with a vacuum and with air in the beam path have been investigated. The performance is compared with that of a flat graphite monochromator.
The construction of a double-radius Gandolfi X-ray camera for the fast generation of well resolved powder-like diffraction patterns of very small single crystals is presented.
Measuring the shift of a pair of properly selected Bragg reflections by double-crystal diffractometry can eliminate the effect of macroscopic, but inhomogeneous, curvature on the strain measurement in single-crystalline layers.
A new algorithm for automatically labelling peaks of an electron density map is described. The procedure has been integrated into the package EXPO.
A new Larmor precession device, consisting of two separate precession modules, operates as a high-resolution neutron diffractometer, without the need for a highly collimated beam, necessary in conventional high-resolution neutron diffraction. The same instrument operates in different modes as an effective small-angle scattering device or accurately determines the line profile, the lattice plane orientation or the angular spread in crystal orientations.
A high-performance neutron diffractometer for biological crystallography (BIX-3) has been constructed. Diffraction data have been collected from the proteins rubredoxin and myoglobin in about one month, to a resolution of 1.5 Å, and were good enough to identify the hydrogen atoms with high accuracy.
A high-intensity two-crystal four-220-reflection germanium monochromator is proposed for high-resolution X-ray diffraction. The beam divergence in the diffraction plane and the fractional wavelength band-pass are smaller by 40% than those of the well known Bartels monochromator, while the flux collected from the source is larger by a factor of five.
A description is given of a method to optimize the design of a time-of-flight diffractometer for the measurement of lattice parameters, based on the identification of a figure of merit to describe the performance of the instrument. Similar figures of merit may be found for other peak shape parameters, and hence for other types of instrumentation.
A novel method to deconvolute the instrumental aberration functions from the experimental powder X-ray data has been developed. The effects of axial divergence, flat specimen, sample transparency and spectroscopic profile of the source X-ray are eliminated from the entire observed diffraction pattern in three-step operations.
A new X-ray scattering process is described, analogous to three-beam diffraction in a single crystal, but with features in the azimuthal scans that are provided by superlattice satellite reflections instead of bulk reflections. These features were named effective satellites and they are observed over the ordinary satellite reflections as a function of the azimuthal angle.
A numerical procedure is proposed for the determination of particle size distributions from neutron double-crystal diffractometer scattering curves influenced by multiple scattering. This procedure is based on an n-fold convolution of single-scattering cross sections, where n depends on the degree of multiple scattering, and makes use of the indirect Fourier transformation method; thus, firstly, size distributions of arbitrary shape can be determined, and secondly, the calculation is fast, allowing an interactive analysis.
Diffraction of high-energy synchrotron radiation with wavelengths of ∼0.1 Å is used to measure the orientation distribution of crystallites in polycrystalline materials. Compared with conventional texture analysis, much higher angular and spatial resolution is reached together with penetration depths in the sample comparable with those of neutrons.
An interpretation of the most recent experimental data on the optical rotatory dispersion of tellurium is presented.
A method is presented that enables the simultaneous determination of the specimen displacement and the temperature of the sample. The method splits the determination of the aberrations into two steps: at room temperature and at elevated temperature.
Download citation


Download citation


Along with the determination of the crystal structure of KYb(WO4)2, a complete description of the single-crystal growth technique is presented and a qualitative morphological study of the crystals grown is described. The thermal linear expansion tensor at room temperature and the optical tensor at λ = 632.8 nm at room temperature are determined.
A procedure is presented for experiments on naked unfrozen protein crystals with the crystal mounted in a conventional cryo-loop and surrounded by a stream of a wet gas. The arrangement allows (i) for rapidly testing crystals for diffraction before freezing, (ii) for data collection between 268–303 K with greatly reduced background, (iii) for the controlled drying or wetting of crystals, (iv) for the anaerobic manipulation of protein crystals, and (v) for the introduction of gaseous or volatile ingredients and reactants into the crystal.
cryocrystallography papers
It is shown that protein crystals may be derivatized with xenon using standard pressure cells when paraffin oil or Panjelly are used instead of conventional cryoprotectant solutions.
short communications
An incommensurate phases database, available on the Internet, is presented.
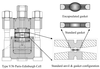
A straightforward modification of the gaskets used in the Paris–Edinburgh pressure cell now permits near-hydrostatic sample compression to pressures of ∼9 GPa. Neutron powder diffraction data collected as part of the initial test programme are presented and discussed, as are possible future developments of sample encapsulation.
computer programs
A library of reusable software components for crystallographic calculations is presented.
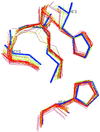
New and improved software tools are described for recognition of protein folds and structural motifs in large databases of protein structures. The tools are fast, sensitive and can be accessed through a user-friendly web-based interface.
laboratory notes
This note focuses on two different ways of enhancing the use of gels in protein crystallization by applying oils to the trials.
crystallographers
Free 

new commercial products
book reviews
Free 

international union of crystallography
Free 



 journal menu
journal menu