issue contents
July 1999 issue
Synchrotron Radiation and Structural Biology

Cover illustration: A schematic representation of the structure of F1-ATPase and Sir John Walker's prize-winning team, see Hasnain, Helliwell and Kamitsubo, pages 809-812.
facility information
editorial
Free 

research papers
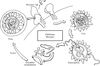
Synchrotron radiation is critically important to the determination of virus structures. Methods of data reduction and phase determination are explained. Some examples are mentioned.
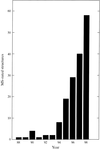
Two decades of synchrotron radiation source, instrumentation and methods evolution have greatly accelerated the pace of protein crystallography, which can, via the linking up of synchrotron radiation `PX stations' worldwide, now yield a `genome level' of numbers of experimentally determined protein structures.
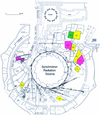
The article is a review of developments in de novo phasing methods and instrumentation in macromolecular crystallography carried out at LURE, the French synchrotron radiation facility.
The course of developments in methodology for multiwavelength anomalous diffraction (MAD) is discussed, and characteristic traits of current applications are analyzed.
XAFS provides local structure of the metal site at subatomic resolution and is equally applicable to both aqueous and crystalline samples. This review provides some recent case studies to illustrate its synergy with protein X-ray crystallographic determination. This synergy exists not only for the scientific objective of structure-based understanding of proteins' function but extends to the source and general instrument requirements, particularly with MAD.
Observations from the structure determination of the bluetongue virus core are presented. Aspects of synchrotron radiation which enable the successful analysis of such complex systems are discussed.
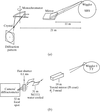
Recent X-ray diffraction/scattering studies on the actomyosin motor system are described and future challenges for synchrotron radiation are discussed.
Major developments in the theory of the Laue diffraction technique and of the necessary software, hardware and experimental strategies have led to successful time-resolved studies on the dynamic properties of a range of biological systems, which shed light on future applications of this technique.
The structure-function relationships of integral membrane proteins so far determined by X-ray and electron crystallographic methods are described.
The MIR map pf the small ribosomal subunit, constructed at 7.2 Å and extended to 5.5 Å using crystals diffracting to 3 Å, shows morphologies close to the active conformation of these particles and contains elongated dense features traceable as RNA chains as well as globular regions in which folds observed in isolated ribosomal proteins were detected. Surface proteins and selected RNA strands are being located by difference Fourier methods, exploiting the metal cluster attached to them.
notes and news
Free 



 journal menu
journal menu