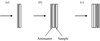issue contents
July 2005 issue
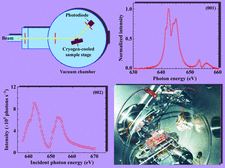
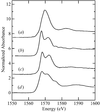
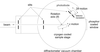
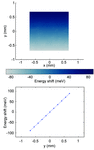
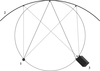
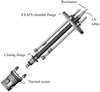
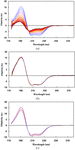
Cover illustration: An in vacuo diffractometer (schematic and apparatus) and the (001) magnetic reflection (top right) and the (002) Bragg reflection for LaSr2Mn2O7 measured with soft X-rays of 600-700 eV (see Hatton, Wilkins, Beale, Johal, Prabhakaran and Boothroyd, pages 434-441), in a collaboration involving the University of Durham, UK; the Institute for Transuranium Elements, Eggenstein-Leopoldshafen, Germany; ESRF, Grenoble, France; CCLRC Daresbury Laboratory, UK; and the University of Oxford, UK.
facility information
softer X-rays
Open  access
access
 access
accessThe application of softer X-ray absorption spectroscopy (XAS) to biological systems is discussed. In addition, the issues of sample preparation, available beamlines, detection devices and other difficulties associated with conducting softer XAS experiments on dilute biological samples are discussed.
With purple membrane the resonant atomic form factors f′ and f′′ of phosphorus vary by nearly 20 electron units at wavelengths near its K-absorption edge at λ = 5.77 Å. Test experiments on a single crystal of ADP/ATP carrier protein using the same wavelength gave a first low-resolution diffraction pattern.
Open  access
access
 access
accessThe utilization and the potential of softer and soft X-rays in macromolecular crystallography as well as the challenges associated with the corresponding diffraction experiments and their possible remedies are reviewed.
Open  access
access
 access
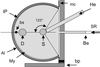
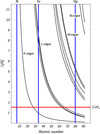
accessThe research effort on novel open-pore chemical structures requires site-specific position and occupancy determination of metal atoms. These involve K edges in the softer X-ray range, from 1.5 to 2.5 Å, leading to novel challenges in synchrotron radiation instrumentation and anomalous dispersion methods in single crystal and powder diffraction. For the latter, separating out of the powder pattern peaks, using softer X-rays, opens up application to ab initio structure solution of more complicated and larger structures.
Open  access
access
 access
accessThe practical aspects of studying powder samples with softer X-rays for indexing or structure solution applications are discussed.
Open  access
access
 access
accessRecent advances in the new technique of soft X-ray single-crystal diffraction are described.
research papers
A novel synchrotron radiation source is proposed that utilizes bulk-type high-temperature superconductors as permanent magnets.
A new Johann spectrometer for high-resolution spectroscopy, which is installed at the wiggler beamline W1 at HASYLAB/DESY, is described. Performance tests and two benchmark experiments (RIXS, XAFS) are presented.
Open  access
access
 access
accessA new MAD beamline at the SRS (UK) for structural genomics and proteomics has been recently commissioned. The beamline is based on a 20 mm external gap, 2.46 T ten-pole wiggler with highly optimized optics to provide rapid (subsecond) tunability over an edge and the highest intensity radiation over 5 to 13.5 keV. An energy-dispersive high-count-rate fluorescence detector with large active area is also integrated into the crystallographic set-up providing unique capabilities to this MAD station.
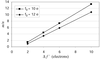
The cube-size contribution to the resolution function of a diced analyzer crystal has been eliminated by the use of a position-sensitive detector. A resolution of 23 meV at a photon energy of 9.9 keV was achieved with a spectrometer based on a 1 m Rowland circle and a diced Si(555) analyzer crystal at a Bragg angle of 88.5°.
The successful fabrication of spherically bent Si analyzer crystals for high-resolution inelastic scattering experiments at transition metal K-edges is reported.
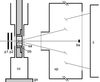
A system for recording micro- and conventional X-ray diffraction patterns from quick-frozen hydrated biological specimens is described. By using this system, diffraction patterns have been obtained from a single sarcomere (volume ∼10 µm3) of an insect flight muscle myofibril.
X-ray diffraction effects from crossed cylindrical surfaces are described. The method is useful for studying contacts between surfaces, liquid confinement and coherence of X-ray beams.
The random-distribution and cluster-formation models for light-induced changes in crystals lead to very different intensity changes and can be distinguished by analysis of the diffraction response to illumination. The effect of light-induced changes on the atomic displacement parameters is analyzed.
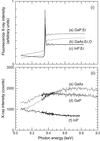
The lowest limit for detection of the impurity concentration doped in semiconductors in the case of fluorescence XAFS measurements has been investigated as a function of the matrix of the impurity and the geometry of the measurement.
A new in situ cell for XAFS measurement in transmission and fluorescence mode is described. As a test case, the oxidation of CO promoted by the Pt/ceria-zirconia catalyst is reported.
An improved normalization algorithm for X-ray absorption spectra is described.
The molecular mechanisms of the protective effect of bisphosphonate drug have been studied by the total reflection X-ray fluorescence method. A molecular film of the membrane enzyme Ca-ATPase deposited on a silicon substrate was used as a model system.
Open  access
access
 access
accessThe effects of very intense synchrotron radiation vacuum ultraviolet light on the circular dichroism spectra and chemical integrity of proteins are discussed. Experiments have been chosen to distinguish between possible degradation and denaturation effects; local heating of the sample resulting in denaturation appears to be the primary factor producing the observed spectral changes.
Open  access
access
 access
accessMethods are described by which the deterioration of the synchrotron radiation circular dichroism signal of proteins in high-flux beamlines can be minimized.
Radiation damage in high-Tc superconductor materials irradiated by high-energy heavy ions has been studied by X-ray fluorescence holography with synchrotron radiation.
short communications
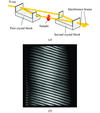
The capabilities of a large-area imaging system based on a two-crystal X-ray interferometer have been extended by raising the X-ray energy from 17.7 keV to 35 keV. An interference pattern and example of a high-quality three-dimensional image are given.
computer programs
A description of a package of graphical data-analysis tools for X-ray absorption spectroscopy is given.
addenda and errata
Free 

current events
Free 



 journal menu
journal menu