issue contents
April 2015 issue
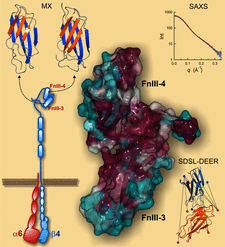
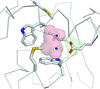
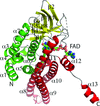
Cover illustration: The structure of the FnIII-3,4 region of the integrin ![[beta]](/logos/entities/beta_rmgif.gif) 4 subunit cytodomain solved by hybrid methods (Alonso-García et al., p. 969). The structures of the individual FnIII domains were solved by macromolecular crystallography (MX). Small-angle X-ray scattering (SAXS) was combined with structure-based site-directed spin labelling (SDSL) and double electron-electron resonance (DEER) to elucidate the relative organization of the two FnIII domains. The structure revealed an evolutionary conserved surface (coloured in purple) that could correspond to a functional site.
4 subunit cytodomain solved by hybrid methods (Alonso-García et al., p. 969). The structures of the individual FnIII domains were solved by macromolecular crystallography (MX). Small-angle X-ray scattering (SAXS) was combined with structure-based site-directed spin labelling (SDSL) and double electron-electron resonance (DEER) to elucidate the relative organization of the two FnIII domains. The structure revealed an evolutionary conserved surface (coloured in purple) that could correspond to a functional site.
research papers
Open  access
access
 access
accessThe crystal structure of hen egg-white lysozyme (HEWL) was analyzed under pressures of up to 950 MPa. The high pressure modified the conformation of the molecule and induced a novel phase transition in the tetragonal crystal of HEWL.
The crystal structure of N-myristoyl transferase from Aspergillus fumigatus complexed with a synthetic inhibitor provides a insight into its selectivity among species.
A crystal structure and biochemical analyses reveal the presence of a PWI-like domain in the N-terminal helicase-associated region of the spliceosomal Brr2 protein, which does not stably bind nucleic acids but instead constitutes part of a protein–protein interaction region that binds several spliceosomal proteins in a yeast two-hybrid assay.
PDB reference: PWI-like domain of Brr2, 4rvq
A series of diffraction images of thaumatin crystals at identical small, 2° rotation intervals were recorded at three different X-ray energies (6.33, 12.66 and 19.0 keV) using five crystals at each energy. The decay in the average diffraction intensities at 100 K owing to radiation damage was analyzed and the dose that diminished the reflection intensities to 70% was determined to be about 7.5 MGy at 6.33 keV and about 11 MGy at the two higher energies.
The L-shaped arrangement of the tandem domains of a double-domain phosphagen kinase exhibits an asymmetric interaction pattern between them. Instead of being linked by a flexible hinge as previously assumed, the two domains are generally fixed with respect to each other, allowing the possibility of inter-domain structural communication.
The p38 MAP kinases are an important family of drug targets for a myriad of inflammatory, as well as other, diseases. Presented here is the crystal structure of the active form of MAPK13 (p38d) as well as a comprehensive analysis of all known apo inactive and active p38 MAPK structures, revealing a common mode of activation as well as some unique structural features.
PDB reference: MAPK13/pTpY, 4myg
The crystal structure of Rubisco activase from A. thaliana is presented. The structure shows a high degree of conformational flexibility and indicates that subtle structural differences may influence interactions in the oligomeric complexes of green-type Rubisco activases.
PDB reference: Rubisco activase, 4w5w
The crystal structure of struthiocalcin-1 (SCA-1) from ostrich eggshell has been determined in two different crystal forms at 1.50 Å resolution, displaying the characteristic C-type lectin-like fold with a surface decorated with acidic residues, highlighting the overall negative charge of SCA-1.
The primary focus of the present work is the study of the effects that two ligands and the crystallization pH have on the crystalline forms of human insulin.
Open  access
access
 access
accessHyp-1, a pathogenesis-related class 10 (PR-10) protein from H. perforatum, was crystallized in complex with the fluorescent probe 8-anilino-1-naphthalene sulfonate (ANS). The asymmetric unit of the tetartohedrally twinned crystal contains 28 copies of the protein arranged in columns with noncrystallographic sevenfold translational symmetry and with additional pseudotetragonal rotational NCS.
Open  access
access
 access
accessExpansion is a modular protein that is conserved in protostomes. The first structure of the N-terminal domain of Expansion has been determined at 1.6 Å resolution and the new Nα-MH2 domain was found to belong to the Smad/FHA superfamily of structures.
PDB reference: Expansion Nα-MH2, 4r9p
The structure of copper type 2-depleted (T2D) laccase from Coriolopsis caperata was solved at 1.6 Å resolution.
The structure of MbtN (Rv1346) presented here supports its annotation as the double-bond-forming dehydrogenase involved in the biosynthesis of mycobactin T in M. tuberculosis and reveals a novel FAD–PEG covalent adduct bound in a unique acyl-binding cavity.
PDB reference: MbtN, 4m6z
The first structure of an A–PCP didomain in which the A domain is in the adenylated state complexed with the intermediate L-Phe-AMP is described.
PDB reference: McyG A–PCP didomain complexed with L-Phe-AMP, 4r0m
Open  access
access
 access
accessThe aggregation process and the fibril state of an amyloidogenic peptide suggest monomer addition to be the prevailing mechanism of elongation and a model of the peptide packing in the fibrils has been obtained.
A Fab fragment with nanomolar affinity for the peptide sequence EYMPME was engineered and characterized by structural, biophysical and computational methods for potential use as a membrane-protein crystallization chaperone.
PDB reference: engineered Fab fragment, 4x0k
Elucidation of the first bacterial chalcone isomerase as native and substrate-bound crystal structures reveals structural homology to a chlorite dismutase, but no structural relation to a plant chalcone isomerase, suggesting a different evolutionary history. The catalytic site of the bacterial isomerase depends on a histidine side chain, in contrast to the catalytic water molecule of the plant enzyme.
PDB references: bacterial chalcone isomerase, 3zph; 4c9s; SeMet derivative, 4c9t; naringenin complex, 4d06
Crystal structures of complexes of type 5 17β-hydroxysteroid dehydrogenase with structurally diverse inhibitors reveal novel scaffolds and conformational changes.
Download citation


Download citation


Open  access
access
 access
accessA microfluidic platform has been developed for the capture and X-ray analysis of protein microcrystals, affording a means to improve the efficiency of XFEL and synchrotron experiments.
PDB reference: hen egg-white lysozyme, 4wmg
A series of high-resolution crystal structures of R. catesbeiana ferritin M loaded with Fe2+ substrate at different metal-ion diffusion times show a sequence of iron-binding sites that allow the visualization of a possible path of substrate diffusion to the catalytic centre.
Cholesterol oxidase: ultrahigh-resolution crystal structure and multipolar atom model-based analysis
The hydrogen-bond stereochemistry and topology of the crystal structure of the flavoenzyme cholesterol oxidase are analyzed at 0.94 Å optical resolution.
PDB reference: cholesterol oxidase, 4rek
Open  access
access
 access
accessThe structure of the FnIII-3,4 region of integrin β4 was solved using a hybrid approach that combines crystallographic structures, SAXS, DEER and molecular modelling. The structure helps in understanding how integrin β4 might bind to other hemidesmosomal proteins and mediate signalling.
Open  access
access
 access
accessStructural and functional characterization of the TesB thioesterase from Y. pestis reveals unique elements within the protomer and quaternary arrangements of hotdog domains, presenting distinguishing features of this enzyme family.
Open  access
access
 access
accessNew structures of the N-terminal 43 kDa fragment of the E. coli DNA gyrase B subunit reveal two discrete monovalent cation-binding sites that could have functional roles.
Open  access
access
 access
accessStructures of the hydrolase domain of 10-formyltetrahydrofolate dehydrogenase from zebrafish and its complexes are reported.
addenda and errata
Open  access
access
 access
access

 journal menu
journal menu