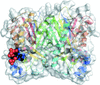issue contents
December 2017 issue
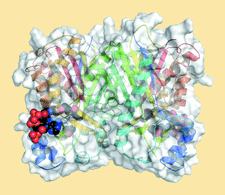
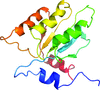
Cover illustration: Biphenyl synthase and benzophenone synthase constitute an evolutionarily distinct clade of type III polyketide synthases that use benzoic acid derived substrates to produce defense metabolites in plants such as apples and pears. Shown here is the structure of biphenyl synthase (BIS) complexed with benzoyl-CoA (Stewart et al., p. 1007). The surface of the protein dimer is displayed and each monomeric subunit is shown in ribbon representation with rainbow colouring from the N-terminus (blue) to the C-terminus (red). Benzoyl-CoA is displayed as spheres with atoms colour-coded according to element: carbon, black; oxygen, red; phosphorus, orange; nitrogen, blue; sulfur, yellow.
research papers
Open  access
access
 access
accessThe yield of protein refolding is reciprocal to the number of proline residues in the target protein. Hydrogen bonds are the primary driving force for de novo protein folding
PDB reference: twinned human AID, 5w09
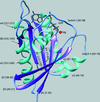
The switch I region in G12A K-Ras undergoes a significant reorganization that results in new interactions between GTP and Tyr32 that stabilize the precatalytic state. These new interactions cause the reduced intrinsic GTP-hydrolysis rate of the G12A K-Ras mutant.
Open  access
access
 access
accessHomology-independent methods for ab initio phasing of α-helical transmembrane proteins are explored.
Open  access
access
 access
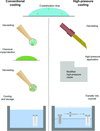
accessA standardized technique for high-pressure cooling of protein crystals has been developed that encompasses all steps from crystal retrieval to automated mounting of the sample at the synchrotron. A wide chemical space for sample cooling has been tested. Amorphous ice was formed in more than 89% of the solutions.
Open  access
access
 access
accessThe structures of biphenyl synthase, of biphenyl synthase complexed with benzoyl-CoA and of benzophenone synthase are compared with that of a chalcone synthase homolog. These structures reveal that benzoic acid-specific type III polyketide synthases contain distinct structural elements, including a novel pocket, which underlie their evolutionary emergence.
An interaction-based fragmentation scheme for solving the issue of scalability in quantum refinement has been developed, implemented in the Q|R software and validated on an amyloid cross-β spine (PDB entry 2oNA).
addenda and errata
Open  access
access
 access
accessbook reviews
Free 



 journal menu
journal menu