issue contents
September 2018 issue
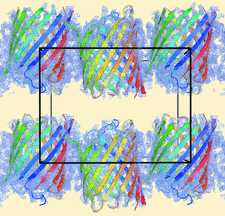
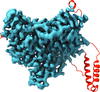
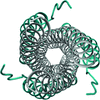
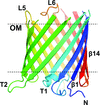
Cover illustration: Crystal packing of the Acinetobacter baumannii outer membrane protein Omp33 (Abellón-Ruiz et al., p. 852). A. baumannii is becoming a major threat to human health as it develops resistance to drugs. This is owing in a large part to the low permeability of its outer membrane, which prevents high internal antibiotic concentrations and makes antibiotic-resistance mechanisms more effective. The structure of one abundant membrane protein, Omp33, helps in understanding how A. baumannii membrane proteins contribute to the low permeability of the cell envelope. Structural information will help us to exploit membrane channels as potential delivery vehicles for future antibiotics.
research papers
Open  access
access
 access
accessNew methods and PHENIX tools for quality assessment of cryo-EM maps, atomic models and model-to-map fitting are presented. Results of systematic application of these tools to high-resolution cryo-EM maps and corresponding atomic models are analyzed and discussed.
Open  access
access
 access
accessThe complete post-fusion core structure of the Human coronavirus 229E spike protein was determined at 1.86 Å resolution. Comparison of the interactions between heptad repeats HR1 and HR2 in different human coronaviruses reveals some differences, which should be taken into consideration when designing pan-coronavirus HR2-mimicking inhibitors that target HR1.
PDB reference: HCoV-229E fusion core, 5yl9
The X-ray crystal structure of Omp33 is reported at a resolution of 2.1 Å. The structure helps in understanding how Acinetobacter baumannii outer membrane proteins contribute to the low permeability of the cell envelope and suggests that Omp33 may function as a ligand-gated channel.
PDB reference: Omp33, 5gie
Relationships between the structure and function of a putative drug target for human African trypanosomiasis have been elucidated using X-ray crystallography and geometric simulations. The data presented provide insights into ligand binding and catalysis, giving direction to future rational drug design.
Open  access
access
 access
accessFor XFEL data, simultaneous refinement of multi-panel detector geometry with thousands of crystal models in the program DIALS improves the integrated signal quality and helps to reduce non-isomorphism
Open  access
access
 access
accessHigh-pressure crystal structure analysis of E. coli dihydrofolate reductase (ecDHFR) complexed with folate and NADP+ was performed up to 800 MPa. A pressure-induced phase transition of monoclinic ecDHFR crystals and structural changes closely related to its reaction mechanism were observed.
High-temperature requirement A (HtrA) proteins belong to the serine protease family and possess protease and PDZ domains. HtrA1 is an essential membrane protein in Mycobacterium tuberculosis.
PDB reference: HtrA1 from Mycobacterium tuberculosis, 5zvj
Open  access
access
 access
accessCryosolution thermal contraction is found to have an impact on cryocooled protein and unit-cell volumes and conformations. In some cases, its adjustment can produce higher quality diffraction data.
scientific comment
Attention is brought to the fact that many coordinate sets deposited in the Protein Data Bank are located outside of the unit cells and are sometimes presented in space groups with symmetry lower than the true space group, and that symmetric oligomers are sometimes scattered, making it harder for the users to interpret quaternary structures.


 journal menu
journal menu