issue contents
May 2020 issue
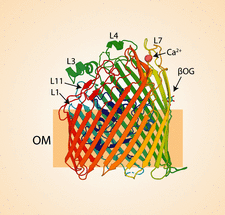
Cover illustration: The crystal structure of TonB-dependent transporter YncD [Grinter & Lithgow (2020), Acta Cryst. D76, 484-495]. In order to transport nutrients across their impermeable outer membrane, Gram-negative bacteria utilize a diverse family of outer-membrane proteins called TonB-dependent transporters. The majority of this family transport iron-containing substrates. However, it is becoming increasingly clear that TonB-dependent transporters target chemically diverse substrates and this article suggests that YncD is unlikely to transport an iron-containing substrate.
letters to the editor
Open  access
access
 access
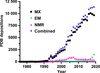
accessFrom the perspective of a young(ish) structural biologist who currently specializes in macromolecular X-ray crystallography, are the best years of crystallography over? Some evidence and hopefully thought-provoking analysis on the subject is presented here.
research papers
The crystal and solution SAXS structures of a fragment of human leucocyte common antigen-related protein show that it is less flexible than the homologous proteins tyrosine phosphatase receptors δ and σ.
Structures of the immunodominant protein P46 from Mycoplasma hyopneumoniae have been determined by X-ray crystallography and it is shown that P46 can bind a diversity of oligosaccharides, particularly xylose, which exhibits a very high affinity for this protein. Structures of a monoclonal antibody that was raised against M. hyopnemoniae cells and specifically recognizes P46 are also reported, both alone and in complex with P46.
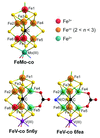
The bond-valence method was performed on 51 crystallographic data sets from nitrogenase proteins, indicating the presence of molybdenum(III) in FeMo cofactors and of vanadium(III) with more reduced iron complements in FeV cofactors.
Death-associated protein kinase 1 (DAPK1) was found to form a complex with purpurin and the crystal structure of the complex was determined. Purpurin may be a good lead compound for the discovery of inhibitors of DAPK1.
PDB reference: DAPK1 in complex with purpurin, 6aar
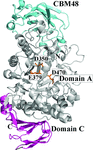
The crystal structures of PbPulA provide the first experimental evidence for functional carbohydrate-binding roles of domain C and CBM48 in GH family 13 pullulanases.
Open  access
access
 access
accessComparison of the structures of ClpC1–ecumicin and ClpC1–rufomycin complexes reveals unique interactions that are relevant to their modes of action.
The apo structure of rice aldo-keto reductase has an ordered open conformation and reveals the key residues that form the substrate-channel wall and determine its substrate preference for straight-chain aldehydes.
PDB reference: AKR4C14, 6kbl
Open  access
access
 access
accessIn this work, the structure and the phylogenetic distribution of the outer-membrane transporter YncD are determined, showing that YncD is unlikely to transport an iron-containing substrate.
PDB reference: outer-membrane transporter YncD, 6v81
This paper reports the structure of Δ1-pyrroline-2-carboxylate reductase from the archaeon T. litoralis, a key enzyme in the second step of trans-4-hydroxy-L-proline metabolism that is conserved in archaea, bacteria and humans.


 journal menu
journal menu