issue contents
November 2020 issue

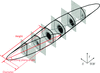
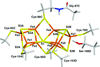
Cover illustration: Lattice-translocation defects in toad neuraminidase [Li et al. (2020), Acta Cryst. D76, 1057-1064]. Neuraminidase (NA) inhibitors are one of the two major classes of antivirals available for the treatment and prevention of influenza. X-ray crystal structure determination of NA head domains and their complexes with various inhibitors are of importance for the design and optimization of anti-influenza drugs. However, the globular tetrameric properties of NA head domains may produce crystals with pathological imperfections, lattice-translocation defects, making structure determination no longer straightforward. Here a crystal of the NA head domain from the Wuhan Asiatic toad influenza virus provides an example of the identification and solution of this type of crystal pathology.
ISDSB2019
research papers


 access
access access
access access
accessbook reviews



 journal menu
journal menu