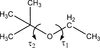issue contents
April 2011 issue
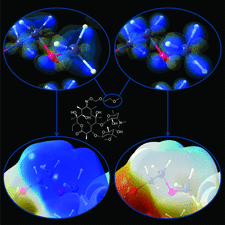
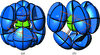
Cover illustration: The cover picture highlights an artefact in the electrostatic potential of the macrolide antibiotic roxithromycin as derived via multipole refinement of X-ray diffraction data. Ignoring the hydrogen disorder (left) in the oxime chain leads to a falsely positive electrostatic potential there as a result of the multipole model's being flexible enough to fit the averaged electron density. If the invariom formalism, which uses theoretical electron density, is applied to the disordered region (right) the artefact disappears. A procedure for obtaining reliable electron density distributions in the presence of disorder has been proposed [Holstein et al. (2010). Acta Cryst. B66, 568-577].
research papers


















 journal menu
journal menu