issue contents
October 2002 issue
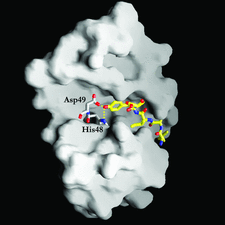
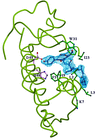
Cover illustration: GRASP (Nicholls et al., 1991) representation of the binding cavity and the hydrophobic channel of DPLA2. The designed peptide LAIYS is almost completely buried in the pocket. Two key hydrogen bonds involving His48 and Asp49 of the protein with peptide Tyr OH are also indicated by dotted lines, p. 1813.
research papers
The X-ray structure of the complex of BW284C51 with T. californica acetylcholinesterase was determined at 2.85 Å resolution and compared with the structures of complexes of the same enzyme with two other active-site gorge-spanning inhibitors, viz decamethonium and E2020.
PDB reference: acetylcholinesterase–BW284C51 complex, 1e3q, r1e3qsf
The application of the SHELXD program to the location of heavy-atom sites from MAD or SAD data is illustrated with the help of examples.
Triplet phases have been obtained for three new proteins from three-beam diffraction. A brief discussion of strategies for the collection of triplet phases and their application in macromolecular crystallography is given.
The structure at 100 K (synchrotron radiation) of the S35C mutant of D. vulgaris flavodoxin at 1.44 Å resolution has been compared with the refined wild-type (wt) structure at 1.34 Å resolution. A different orientation of the Cys35 thiol group with respect to the Ser35 hydroxyl has been found.
An analysis of length and residue distributions, interactions and location in the overall tertiary structure of α-helices and β-strands at polypeptide-chain termini is presented and discussed in the context of protein folding and design.
The crystal structures of two mutants of horseradish peroxidase C (HRPC) with substitutions of the key catalytic residues Arg83 and His42 have been determined to 2.5 Å (H42E HRPC) and 2.0 Å (R38S/H42E HRPC) resolution.
The structure of the complex formed between Russell's viper phospholipase A2 and a designed peptide LAIYS has been determined at 2.0 Å resolution. The peptide binds to phospholipase A2 specifically and fills the hydrophobic channel completely.
PDB reference: PLA2–LAIYS, 1jq8
A fast method for fitting model electron densities into EM reconstructions is presented. This new procedure was inspired by a rigid-body refinement method used in macromolecular X-ray crystallography.
crystallization papers
To investigate how the BRCT domains of 53BP1 protein bind to p53, the 53BP1 BRCT region in complex with the DNA-binding domain of p53 was overexpressed, purified and crystallized. The crystals of the complex belong to the space group P212121 and diffraction data have been collected to 2.6 Å resolution.
The mutant α-subunit of group II chaperonin from Thermococcus strain KS-1 was crystallized. Diffraction data were collected to 2.4 Å with a synchrotron-radiation source.
A plant antifungal protein, Gastrodia antifungal protein (GAFP-1) has been purified from terminal corms of the orchid G. elata and crystallized in space group P21212 and with unit-cell parameters a = 61.087, b = 91.488, c = 81.132 Å. A set of diffraction data to resolution 2 Å has been collected with an overall Rmerge of 0.097 and a completeness of 99.8%, which will help to solve the first structure of a monomeric monocot mannose-binding lectin.
A non-toxic acidic phospholipase A2 from the venom of O. hannah (king cobra) has been crystallized and X-ray data have been collected and reduced to 2.1 Å resolution. Analysis by molecular replacement showed there to be six enzyme molecules per asymmetric unit.
A novel antifungal protein with five disulfide bridges from E. ulmoides Oliver has been crystallized. X-ray diffraction data at 1.0 Å have been collected and used to obtain phase information directly by an ab initio method.
10-Formyltetrahydrofolate dehydrogenase is important for the regulation of intracellular folate pools, for de novo purine biosynthesis and for the removal of formate in the form of CO2. Crystals of the amino-terminal hydrolase domain of this enzyme that diffract to 2.3 Å resolution have been obtained.
NKp44, a natural killer cell activating receptor, is composed of an extracellular region (168 amino acids), a membrane-spanning segment (25 amino acids) and a cytoplasmic domain (63 amino acids). The extracellular region Ig-like domain, held to be involved in target-cell recognition/binding, has been crystallized in a form suitable for structural investigation.
Cyclophilin from the parasite L. donovani has been crystallized, with crystals diffracting up to 3.5 Å resolution. Unambiguous solutions in rotation and translation functions have been obtained with human cyclophilin as the search model.
Human neuroglobin (151 amino acids), a haemoglobin involved in the neuronal response to hypoxia or ischaemia, has been expressed, purified and crystallized as a Cys-free mutant protein, with a view to three-dimensional structure determination.
Dpr, a ferritin-like protein from S. suis, was expressed in E. coli, purified and crystallized. Full data were collected to 2.3 Å resolution using synchrotron radiation.
Crystallization and preliminary diffraction data analysis for recombinant human annexin 2 to 1.52 Å resolution.
Cyclase-associated protein (CAP) from D. discoideum, a protein involved in signal transduction, has been crystallized and a complete native data set has been collected at 2.2 Å resolution.
The DNA-binding domain of the transcriptional activator PhoB (PhoBC) has been expressed in E. coli and crystallized in two crystal forms, one of which led to experimental maps at 3.0 Å resolution.
The multi-drug efflux transporter AcrB from E. coli was purified and crystallized. AcrB crystallizes in space group R32, with unit-cell parameters a = b = 143, c = 513 Å. Crystals diffract to 3.0 Å resolution.
To aid studies in understanding gametocytogenesis in P. falciparum, Pfg27 has been expressed and purified yielding ∼5 mg l−1 of purified protein in soluble form. This protein has been crystallized by the hanging-drop vapor diffusion method using PEG 8000 as a precipitant. Native diffraction data were collected under cryogenic conditions, and phase resolution by selenium-methionine aided multiple-wavelength anomalous dispersion technique is in progress.
A C-terminal 30 kDa fragment of the selenocysteine-specific elongation factor SelB from M. thermoacetica has been recloned, expressed in E. coli and crystallized in a suitable form for X-ray analysis. The crystals diffract to 2.7 Å in an in-house X-ray source.
PDB reference: SelB370–634, 1lva, r1lvasf
Peptide deformylase from P. aeruginosa has been crystallized (P212121; a = 68.75, b = 74.46, c = 77.18 Å). Diffraction data have been collected to 1.85 Å using synchrotron X-rays.
The nucleosome-assembly factor Cia1p from S. cerevisiae has been crystallized by the hanging-drop method. A complete diffraction data set using synchrotron radiation was collected to 2.95 Å resolution.
Crystallization of bucain, a three-finger toxin structurally homologous to snake-venom muscarin toxins, from the venom of the Malayan krait B. candidus is presented.
Mlc1p is a myosin light chain that binds to IQ motifs of a class V myosin (Myo2p) and an IQGAP-like protein (Iqg1p), playing a role in cytokinesis and organelle transport in S. cerevisiae. Complexes of Mlc1p with IQ motifs 2, 3, 4, and 2,3 of Myo2p were crystallized and their structures determined.
Construct variation in conjunction with diffraction data analysis were used in the successful crystallization of the TRAF domain of TRAF6.
short communications
A web-based database-backed crystallographic notebook system is presented.
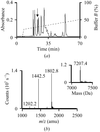
A large volume of electron density in the crystal of Escherichia coli N-acetyl-L-glutamate kinase is interpreted as an external AMPPNP molecule that makes contacts with the enzyme through its γ-imidophosphate. This external nucleotide differs in a number of respects from the internal AMPPNP bound to the active site, and the possibility that the external AMPPNP is waiting to occupy the active center is discussed.


 journal menu
journal menu