issue contents
January 2017 issue
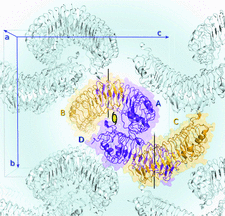
Cover illustration: Arrangement of molecules within the crystallographic unit cell of the human cartilage matrix protein chondroadherin (Rämisch et al., p. 53). Peptides based on the C-terminal human chondroadherin sequence have shown therapeutic potential for treating osteoporosis. The asymmetric unit is shown in colour (chains A, B, C and D); dimers are formed by chains AB and CD. Vertical black lines indicate the intra-dimer twofold symmetry axes, and the second twofold-symmetry axis, relating chains A and D, is indicated in the centre of the asymmetric unit, almost parallel to the a axis of the unit cell.
research papers
A crystal structure of the U6 snRNP core from yeast shows that mutation of the U6 internal stem-loop (ISL) does not alter the overall topology of the snRNP, but suggests structural plasticity in the ISL.
PDB reference: U6–Prp24 complex, 5tf6
X-ray structures of the enzyme 5-aminolaevulinate dehydratase (ALAD) purified from human blood, as well as the recombinant human form and the ALAD from the hyperthermophilic archaeon P. calidifontis, are reported, together with the structure of E. coli ALAD co-crystallized with a noncovalently bound moiety of the product, porphobilinogen. The structures give insight into the zinc-dependence of these enzymes, the possible role of electrostatics in substrate funnelling and details of the interactions made by the bound pyrrole end product.
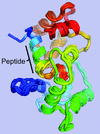
A description is given of the structure solution of a nearly perfectly hemihedrally twinned novel calmodulin–peptide complex by molecular replacement in combination with single-wavelength anomalous diffraction. The resulting structure revealed a unique mode of peptide–calmodulin interaction in which the peptide is bent to nearly 90° within the calmodulin-binding pocket.
PDB reference: a novel peptide–CaM complex, 5dow
The structure of the calcium-dependent phosphoinositide-specific phospholipase C from Pseudomonas sp. 62186 is described, the first from a Gram-negative bacterium. Structure solution from a low-sequence-identity homology model is described in detail.
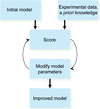
Quantum-based refinement software is being developed to refine biomacromolecules against crystallographic or cryo-electron microscopy data.
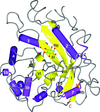
The structure of human chondroadherin (hCHAD), a 359-amino-acid protein with 11 leucine-rich repeats, has been solved by molecular replacement using de novo models of a 195-residue segment and the AMPLE pipeline. This demonstrates that even a fairly large β-rich protein is tractable to solution using de novo modelling. The structure of the C-terminal domain of hCHAD reveals the conformation of a medically relevant peptide.
PDB reference: human chondroadherin, 5lfn
In the course of soaking experiments of lytic polysaccharide monooxygenase crystals with potential oligosaccharide ligands, local changes in crystal packing, various degrees of active-site disorder and binding of oligosaccharides at potential branching or surface binding sites were observed.
PDB references: Zn-AoAA13, 5t7j; AoAA13-His91flip, 5t7k; AoAA13-G2(α1,4), 5lsv; AoAA13-G3(α1,4)G1(α1,6), 5t7n


 journal menu
journal menu