issue contents
November 2019 issue
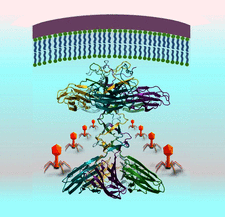
Cover illustration: The Bacillus subtilis PBSX prophage lytic cassette protein XepA adopts a dumbbell shaped pentamer. Structural analysis suggests that XepA may interact with the cytoplasmic membrane to facilitate cell lysis [Freitag-Pohl et al. (2019), Acta Cryst. D75, 1028-1039].
research papers
Open  access
access
 access
accessHigh-energy phase-contrast X-ray microscopy and tomography of protein crystals in an optically opaque matrix is demonstrated with micrometre resolution on the macromolecular crystallography beamline P14 at PETRA III.
Open  access
access
 access
accessSAD phasing of XFEL data is shown to require accurate error estimates from the merged reflection intensities. Various methods of treating the errors are presented, including a best-practice approach that refines error-correction terms using a nonlinear least-squares method.
The Sonic Hedgehog (Shh) protein is essential for embryonic patterning, but can contribute to various cancers when the underlying signaling pathway is aberrantly activated. Here, the structure of the human Shh signaling domain is presented in complex with octahedrally coordinated Zn2+ and Mg2+ ions at 1.43 Å resolution that give rise to localized changes. The modes of ion coordination are compared with previous structures determined in isolation or in complex with binding partners, which reveals significant plasticity in the modes of metal coordination that has implications for the design of peptide drugs to repress the signaling pathway.
PDB reference: human Sonic Hedgehog N-terminal signaling domain, 6pjv
By maintaining internal solvent in a fully liquid state, temperature-dependent and time-dependent crystal disordering and reordering, protein conformational relaxations and clear evidence for solvent flows following cooling can be observed in apoferritin crystals at temperatures between 220 and 260 K. These results illuminate the causes of and remedies for cooling-induced crystal disorder, and suggest the feasibility of studying aspects of cold denaturation under more nearly native solvent conditions at temperatures down to 200 K.
Open  access
access
 access
accessThe structure of AtzT, which is thought to be involved in atrazine uptake in bacteria, was solved by SAD phasing using an ethylmercury phosphate derivative. Density in the binding site was subsequently found to be guanine that was retained by the protein throughout the purification process. Replacing the guanine with 2-hydroxyatrazine allowed the manner of binding of this substrate to be determined and its comparison with what appears to be the original ligand for this protein.
Open  access
access
 access
accessMcl-1 is an important cancer target for drug therapy, through which normal apoptosis may be restored by inhibiting its protective function. An scFv and a Fab have been used to generate an apo Mcl-1 crystal system that is amenable to iterative structure-guided drug design.
The nature of crystallography of biological macromolecules can be unpredictable, and protein crystals do not consistently produce high-resolution diffraction data. In the study described here, it was sought to develop a computational design approach that could identify resolution-enhancing point mutations at crystallographic protein–protein interfaces.
Open  access
access
 access
accessThe lytic cassette proteins XepA and YomS from Bacillus subtilis prophages have been characterized and it was found that only XepA establishes cytotoxic activity in plaque assays. The crystal structures of both proteins show a unique pentameric assembly, in which YomS adopts a very similar fold to the C-terminal domain of the XepA dumbbell pentamer. The overall architecture of XepA, with the N-terminal domain subunits resembling cytoplasmic membrane-binding C2-domain folds, suggests that any lytic functionality could be based on disruption of the proton motive force of the cytoplasmic membrane, which induces cell lysis.


 journal menu
journal menu